- Home
- News
- Spotlight on Science
- Record paramagnetic...
Record paramagnetic response of gold nanoparticles deposited on an archaeal cell wall surface layer
31-01-2013
Gold nanoparticles were grown on the surface layer (S-layer) of the outer cell wall of the thermoacidophilic archaeon Sulfolobus acidocaldarius. Cysteine residues in the cell wall provide sulphur atoms that render the Au paramagnetic by increasing the number of holes in its 5d band. The paramagnetic response of the nanopaticles to an external field was measured using XMCD at the Au L2,3 absorption edges.
For millennia, gold has been used by humanity as an exchange token, and also as an element on which to express art and beauty because of its inertness and brilliance. However, when the size of a gold sample is reduced to the nanometre scale, its physical properties change. In bulk form, gold is a diamagnetic metal, while nanoparticles of diameter 1.4 nm, capped with thiol molecules, show a paramagnetic response, albeit a very small one [1]. During the last decade, there has been a strong debate on whether this magnetism is intrinsic to the Au atoms forming the nanoparticle, or whether it arises from extrinsic effects at the surface, impurities, or other possible sources of a minute magnetic signal.
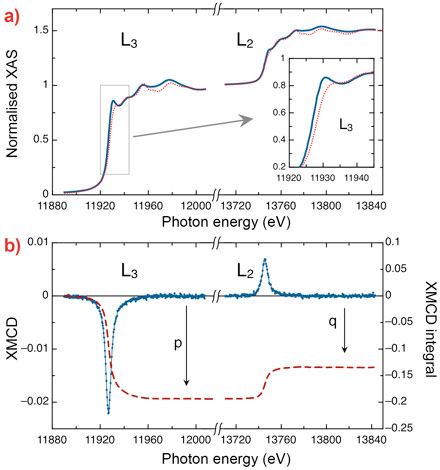
X-ray magnetic circular dichroism (XMCD) measured at the Au L2,3 edges is an element-selective technique. A few XMCD studies have already been performed on Au-thiol capped nanoparticles, permitting the magnetism of 5d origin to be observed. However, in these experiments, the signals were minute, barely above experimental uncertainties [2].
The samples studied in our experiments were produced at the HZDR. We detected the highest paramagnetic response ever observed on gold atoms by using XMCD. It was possible to demonstrate that Au is at the origin of the observed magnetic moments by XMCD. The dominant signal corresponded to the 2p → 5d electron excitation transitions, thus we probed the magnetism due to the 5d band of Au. This observed response was 25 times larger than any measured before for nanoparticles (Figure 1). This achievement was possible thanks to the unique combination of a 17 T magnetic field and 2 K temperature, conditions that were necessary for this experiment and only available at beamline ID12. This work has been completed with electron microscopy at the University of Granada and the Laboratorio de Microscopías Avanzadas (LMA) at the Instituto de Nanociencia de Aragón (INA) in Zaragoza, and ultralow temperature magnetometry at ICMA.
The biological template used for synthesis of the Au nanoparticles consisted of empty microbial cells (“ghosts”), composed of the extremely-stable, highly-ordered and porous proteinaceous S-layer of the thermoacidophilic archaeon Sulfolobus acidocaldarius. This archaeon thrives in sulphur-rich ponds such as those in Yellowstone National Park. The monomers of this S-layer protein contain two cysteine amino acid residues, each with a C-S-H thiol side chain. The production of the archaeal Au nanoparticles was performed in a two-step procedure. The first step consisted of Au(III) deposition onto the S-layer, achieved by incubation of the “ghosts” in a solution of HAuCl4. The second step, performed after the removal of the non-deposited Au(III), comprised the chemical reduction of Au(III) into metallic Au(0), forming the Au nanoparticles on the S-layer template [3]. The average size of the nanoparticles was 2.6 nm wide.
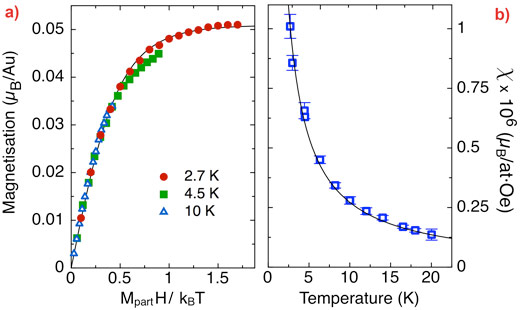
Both XAS and XMCD measurements on Au L2,3 and S K edges were carried out at beamline ID12. The XAS data show that Au exchanges electrical charge with the biological matrix since the number of holes in the 5d band increases with respect to the bulk Au. The exchange takes place via Au binding with sulphur of the S-layer template. From the Au XMCD data, the evolution of the magnetisation as a function of applied field was determined up to 17 T (Figure 2a), and found to obey Langevin’s equation. The average particle magnetic moment extracted from the data was found to be 2.4 Bohr magnetons, and the average magnetic moment per Au atom 0.055 Bohr magnetons.
The magnetisation depends on temperature and follows the Curie law expected for a paramagnetic particle, this was obtained from the XMCD peak measured at fixed photon energy (Figure 2b). Micro-SQUID ac susceptibility measurements performed with a 3He-4He refrigerator show that this paramagnetic behaviour extends down to at least 0.016 K. The magnetic anisotropy is, therefore, extremely low.
The spins located at the Au 5d band appear to be coupled by spin-spin interactions, and the spin-orbit coupling generates an appreciable orbital moment, which amounts to nearly one third of the total magnetic moment of an average paramagnetic Au atom. The striking magnetic properties of these Au nanoparticles are due to the special synthesis on a unique biological matrix containing a low number of thiol groups distributed in a highly ordered way.
Principal publication and authors
Strong paramagnetism of Au nanoparticles deposited on a Sulfolobus acidocaldarius S-layer, J. Bartolomé (a,b), F. Bartolomé (a,b), L.M. García (a,b), A.I. Figueroa (a,b), A. Repollés (a,b), M.J. Martínez (a), F. Luis (a,b), C. Magén (b,c), S. Selenska-Pobell (d), F. Pobell (d), T. Reitz (d), R. Schönemann (d), T. Herrmannsdörfer (d), M. Merroun (e), A. Geissler (d), F. Wilhelm (f), and A. Rogalev (f), Physical Review Letters 109, 247203 (2012).
(a) Instituto de Ciencia de Materiales de Aragón (ICMA), CSIC - Universidad de Zaragoza (Spain)
(b) Departamento de Física de la Materia Condensada, Universidad de Zaragoza (Spain)
(c) Laboratorio de Microscopías Avanzadas (LMA), Instituto de Nanociencia de Aragón (INA) - ARAID, Universidad de Zaragoza (Spain)
(d) Institute of Resource Ecology and Dresden High Magnetic Field Laboratory, Helmholtz-Zentrum Dresden-Rossendorf (HZDR), Dresden (Germany)
(e) Department of Microbiology, University of Granada (Spain)
(f) ESRF
References
[1] P. Crespo et al., Physical Review Letters 93, 087204 (2004).
[2] Y. Yamamoto et al., Physical Review Letters 93, 116801 (2004).
[3] S. Selenska-Pobell et al., Nanomater and Nanotechnology 1, 8 (2011).
Top image: Gold nanoparticle grown on a Sulfolobus acidocaldarius S-layer imaged by high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM).