- Home
- News
- Spotlight on Science
- Structural disorder...
Structural disorder accelerates the kinetics of electrocatalytic reactions
19-05-2017
Operando experiments at beamline ID31 revealed an important link between structural defects in PtNi/C nanoparticles and an enhancement of the kinetics of the oxygen reduction reaction. Subsequently, by designing structural disorder into the system, a new class of catalyst has been developed with performance comparable to the best shape and composition controlled nanocatalysts but with higher stability.
The electrochemical activation of oxygen is the cornerstone of electrochemical conversion and storage devices such as proton-exchange membrane fuel cells and water electrolysers (PEMFC and PEMWE, respectively) and metal-air batteries. A key element, of both fundamental and applied importance, is the electrochemical reduction of oxygen, the kinetics of which governs the electrical performance of a PEMFC. Platinum is the most active electrocatalyst for the oxygen reduction reaction (ORR) in an acidic electrolyte. Alloying Pt with a transition metal (M) such as Ni, Co or Cu enhances its catalytic performance while minimising the mass of Pt used. The presence of the transition metal modifies the electronic structure of Pt by strain (the Pt-Pt distance is compressed in PtM alloys resulting in a change in the overlap of electronic orbitals) and ligand effects (the neighbouring M atoms exert a direct influence on the electronic structure of Pt), resulting in weakened chemisorption of intermediates and accelerated ORR kinetics. To date, carbon-supported PtM nanoalloys (PtM/C) or core@shell nanoparticles composed of a Pt-enriched shell and a metallic or alloyed core (M@Pt/C or PtM@Pt/C), represent the best industrial compromise between catalytic activity and cost.
To achieve further gains in catalytic activity with minimisation of cost, we are currently developing new catalytic architectures. Porous hollow PtNi/C nanoparticles, composed of a PtNi shell surrounding a nanoscale void are of particular interest. A 4-fold enhancement in specific activity and a 3-fold enhancement in mass activity have recently been obtained for this new family of catalysts compared to solid PtNi/C nanoparticles with similar crystallite size and Ni content. This result was a first indication that, beyond the classical strain and ligand effects, other parameters play a crucial role. Experiments conducted at the ESRF have proved crucial in identifying these parameters and understanding in quantitative terms the peculiar ORR activity of this class of porous nanocatalysts [1].
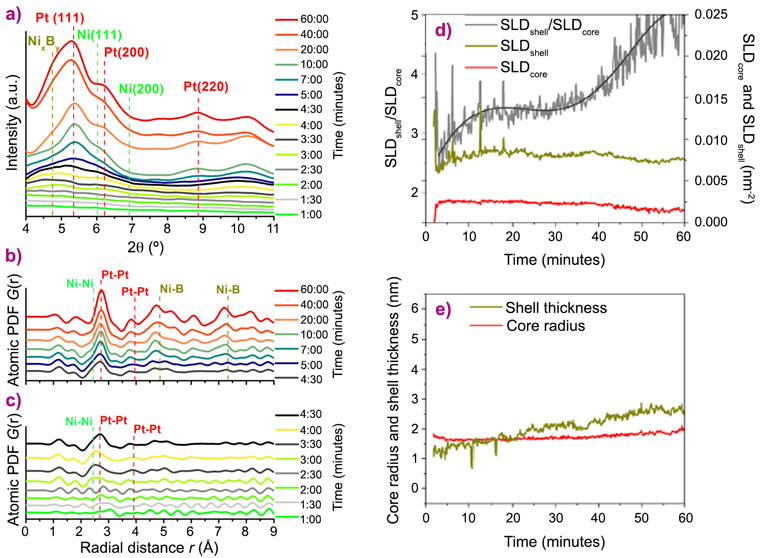
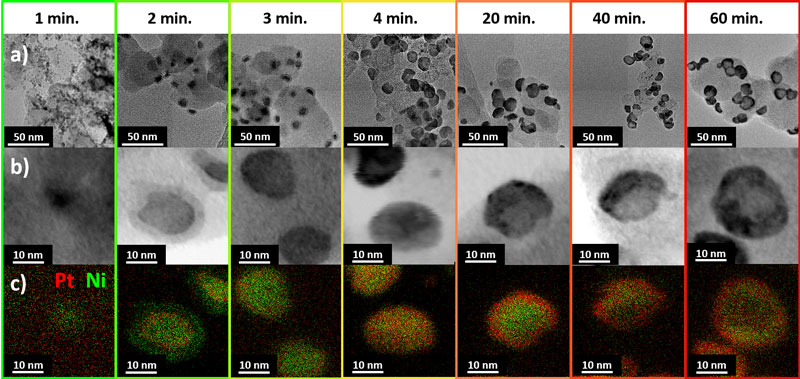
By combining synchrotron small- and wide-angle X-ray scattering (SAXS and WAXS), aberration corrected high-resolution transmission electron microscopy (HR-TEM), scanning transmission electron microscopy (STEM), energy dispersive X-ray spectroscopy (X-EDS) and electrochemical techniques (Figure 1 and Figure 2), we have recently proposed a comprehensive formation and growth mechanism for hollow PtNi/C [2]. Operando SAXS and WAXS experiments performed at beamline ID31 revealed that during the synthesis of hollow PtNi/C nanoparticles, Ni-rich/C nanoparticles form first (0 < t ≤ 1 min), before being embedded in a NixByOz shell 1 < t ≤ 2 min). The combined action of galvanic displacement and of the nanoscale Kirkendall effect then results in the sequential formation of Ni-rich core@Pt-rich/C shell (2 < t ≤ 20 min) and ultimately hollow PtNi/C nanoparticles (20 < t ≤ 60 min), see Figure 1. Moreover, Rietveld refinement of the WAXS patterns revealed that the PtNi shell surrounding the particle central void in the final catalyst is composed of small crystallites (~3 nm) interconnected to each other via grain boundaries.
 |
|
Figure 1. Nanostructures forming during the synthesis of hollow PtNi/C nanoparticles: a) Conventional TEM images; b) STEM images; c) elemental maps. |
The high density of grain boundaries and the presence of atomic vacancies in the final porous hollow PtNi/C nanocatalysts induce a strong atomic disorder, which was quantified with the help of microstrain values [2]. Microstrain, also known as local lattice strain, is a fingerprint of the deviation of atoms from their ideal positions because of structural defects (stacking faults, twins, grain boundaries, and/or dislocation arrays). Using post-synthesis thermal annealing, we were able to show that structural defects present in hollow PtNi/C nanocatalysts positively influence the ORR kinetics.
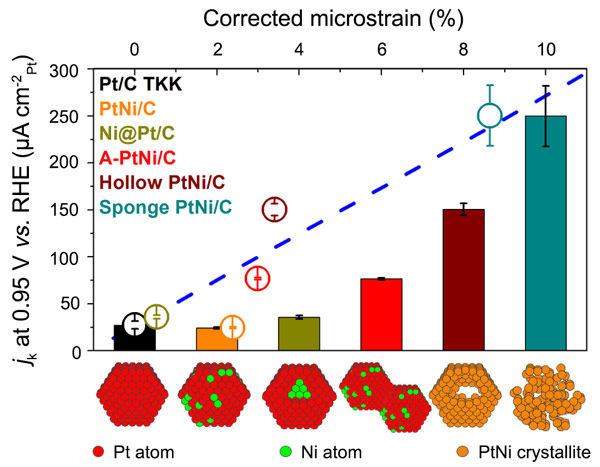
Two approaches were used to verify whether this concept may be extended to other bimetallic nanocatalysts. Firstly, nanometre-sized PtNi/C electrocatalysts with similar Ni content (~15 at. %) but different nanostructures and different fraction of structural defects were synthesised and characterised: solid, hollow or “sea sponge” PtNi/C nanoalloys, Ni@Pt/C core@shell nanoparticles. A 9-fold enhancement of the ORR specific activity was obtained between the worst and the best performing PtNi/C catalysts showing that structural effects outweigh the strain and ligand effects at low Ni at. % (see Figure 3) [3]. Secondly, density functional theory calculations confirmed that highly disordered lattices have peculiar surface reactivity [4].
In conclusion, the key parts of the discovery were the precise quantitative determination of the microstrain values describing local structural disorder and the mastering of the synthesis guided by the operando experiments. This research unequivocally demonstrates that large-scale research facilities such as ESRF play a crucial role in advancing research into energy materials. It also paves the way to improved and sustainable ORR electrocatalytic performance for PEMFC under real operating conditions.
Principal publication and authors
[1] Defects do catalysis: CO monolayer oxidation and oxygen reduction reaction on hollow PtNi/C nanoparticles, L. Dubau (a,b), J. Nelayah (f), S. Moldovan (g), O. Ersen (g), P. Bordet (d,e), J. Drnec (c), T. Asset (a,b), R. Chattot, (a,b) and F. Maillard (a,b), ACS Catal., 6, 4673−4684 (2016); doi: 10.1021/acscatal.6b01106.
[2] Atomic scale snapshots of the growth mechanism of hollow PtNi/C nanocatalysts for oxygen reduction reaction, R. Chattot (a,b), T. Asset (a,b), J. Drnec (c), P. Bordet (d,e), L. Dubau (a,b) and F. Maillard (a,b), Nano Lett. 17, 2447–2453 (2017); doi: 10.1021/acs.nanolett.7b00119.
[3] Beyond strain and ligand effects: microstrain-induced enhancement of the oxygen reduction reaction kinetics on various PtNi/C nanostructures, R. Chattot (a,b), T. Asset (a,b), P. Bordet (d,e), J. Drnec (c), L. Dubau (a,b) and F. Maillard (a,b), ACS Catal. 7, 398-408 (2017); doi: 10.1021/acscatal.6b02356.
(a) Univ. Grenoble Alpes, LEPMI, Grenoble (France)
(b) CNRS, LEPMI, Grenoble (France)
(c) ESRF
(d) Université Grenoble Alpes, Institut Néel, Grenoble (France)
(e) CNRS, Institut Néel, Grenoble (France)
(f) Univ. Paris Diderot, CNRS, LMPQ, Paris (France)
(g) Univ. Strasbourg, IPCMS, Strasbourg (France)
References
[4] Effect of atomic vacancies on the structure and the electrocatalytic activity of Pt-rich/C nanoparticles: a combined experimental and density functional theory study, O. Le Bacq, A. Pasturel, R. Chattot, B. Previdello, J. Nelayah, T. Asset, L. Dubau, F. Maillard, ChemCatChem, (2017); doi: 10.1002/cctc.201601672.