- Home
- News
- Spotlight on Science
- Inflammation increases...
Inflammation increases gadolinium retention in the brain after administration of a linear gadolinium-based contrast agent
12-06-2019
Gadolinium-based contrast agents are used in neuroradiology to monitor diseases such as the neuroinflammatory multiple sclerosis. The processes by which Gd is deposited in tissues remains undetermined. The effect of inflammation on Gd spatial distribution and concentration in brain tissues has been investigated with synchrotron fluorescence spectroscopy at beamlines ID21 and ID16B.
In multiple sclerosis (MS), gadolinium-based contrast agent (GBCA)-enhanced magnetic-resonance imaging is commonly used in disease diagnosis and monitoring. Linear GBCA is a variant in which a long organic molecule wraps around the gadolinium ions without enclosing then completely. In the past years, deposits of Gd were detected in various tissues, principally brain and bones, of patients that had previously received GBCA. However, it remains unclear whether such deposits have consequences for the patient’s well-being, and whether neuroinflammation and blood-brain-barrier disruption (characteristic of MS) affect Gd retention inside the brain. Here, micro- and nano-synchrotron X-ray fluorescence spectroscopy (SR μXRF and SR nano-XRF, respectively) was used to evaluate Gd retention in healthy and inflamed murine tissues that were exposed in vivo to repeated linear GBCA injections (eight injections, cumulated dose of 20 mmol/kg). SR μXRF and SR nano-XRF maps were collected at beamlines ID21 and ID16B.
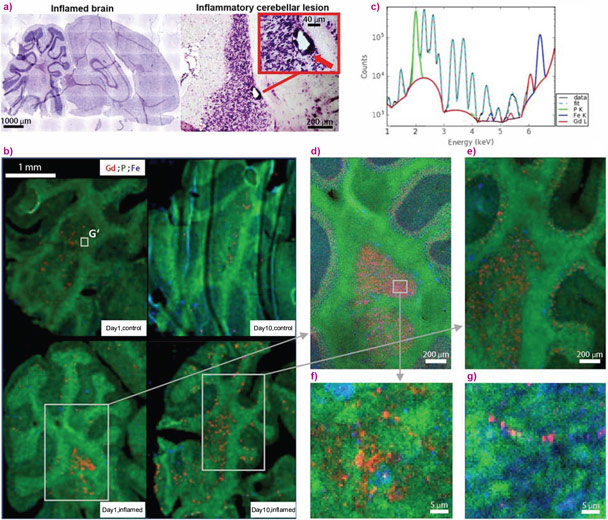
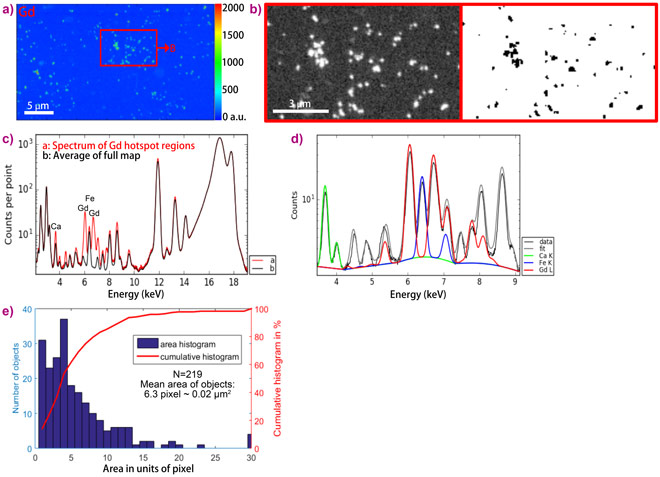
SR-XRF revealed that Gd deposits increased during inflammation and were located in areas of high inflammatory activity. H&E staining was used to identify areas with inflammation (red arrow in Fig. 1a). These areas were then selected for SR-µXRF mapping of tissue obtained one day or ten days after the last GBCA application (Fig. 1b, bottom set of images). Corresponding healthy brain areas are shown in Fig. 1b (top set of images). Large µXRF overview maps indicate that although Gd retention was observed in healthy and inflamed tissue, retention is enhanced in the inflamed cerebellum. Furthermore, the analysis of Gd distribution revealed Gd accumulation in small spots, visible in maps obtained at 3 µm step size (Fig. 1d-e) and even more pronounced at 500 nm step size (Fig. 1f-g), for both healthy and inflamed brain tissue. Although the amount of hotspots is increased in inflammation, Gd accumulated in those spots is very high (up to ~6 mg Gd/g tissue) in both, healthy and inflamed brains. This high concentration is remarkable and may have impact for further risk assessment of GBCA, especially since ICP-MS revealed that the overall tissue concentration of the same samples is only about 1-10 µg/g. Finally, nano-XRF investigation (at 60 nm resolution) of an inflamed brain sample at ID16B suggested an average size of Gd hot-spot regions of ~160 nm diameter, containing around 3000-5000 ppm mass fraction of Gd (Fig. 2).
The finding of this study on Gd concentration, co-localisation with other elements, as well as Gd spatial distribution on a sub-micron length scale are thanks to the unique properties of the XRF-setups installed at the two beamlines used. Though the benefits of using GBCA in clinical routines are evident, a comprehensive risk assessment on the use of linear GBCAs should therefore consider a potentially increased risk of Gd retention, in particular, for patients suffering from chronic neuroinflammatory disorders.
Principal publication and authors
Increased retention of gadolinium in the inflamed brain after repeated administration of gadopentetate dimeglumine: A proof-of-concept study in mice combining ICP-MS and micro- and nano-SR-XRF. S. Wang (a,b), B. Hesse (c,d), M. Roman (e), D. Stier (d), H. Castillo-Michel (c), M. Cotte (c,f), J.P. Suuronen (c), A. Lagrange (d,g), H. Radbruch (h), F. Paul (i), M. Taupitz (j), E. Schellenberger (j), I. Sack (j), C. Infante-Duarte (a), Invest Radiol. (2019); doi: 10.1097/RLI.0000000000000571.
(a) Institute for Medical Immunology, Charité – Universitätsmedizin Berlin (Germany)
(b) Department of Neurology, Xiangya Hospital, Central South University, Changsha (China)
(c) ESRF
(d) Xploraytion GmbH, Berlin (Germany)
(e) European Center for the Sustainable Impact of Nanotechnology, EcamRicert Srl, Padua (Italy)
(f) Laboratoire d’Archéologie Moléculaire et Structurale (LAMS), Sorbonne Université, CNRS, UMR8220 Paris (France)
(g) Materials Engineering, Berlin Institute of Technology (Germany)
(h) Department of Neuropathology, Charité – Universitätsmedizin Berlin (Germany)
(i) NeuroCure Clinical Research Center and Experimental and Clinical Research Center, Charité – Universitätsmedizin Berlin and Max Delbrueck Center for Molecular Medicine, Berlin (Germany)
(j) Department of Radiology, Charité – Universitätsmedizin Berlin (Germany)