- Home
- News
- Spotlight on Science
- Improving the lifetime...
Improving the lifetime of auto exhaust catalysts
16-12-2008
Today there is great motivation to improve vehicle exhaust catalysts because of our increasing appreciation of the damage that toxic gas emissions are doing to the environment in tandem with the finite availability of noble metals. The catalyst lifetime is drastically reduced when the highly-dispersed metal particles agglomerate and sinter, decreasing the active surface area. Recent in situ X-ray studies of Pt catalysts on ceria-based supports have provided an understanding of the processes involved, which will lead to “on board” methods to improve the lifetime of the catalysts fitted to our cars.
Supported precious metals are used to facilitate many industrial catalytic processes. Platinum (Pt) in particular is found at the core of catalysts used for the cleaning up of motor vehicle exhaust emissions. When the exhaust catalyst is exposed to high temperatures (800°C and above), the highly-dispersed metal nanoparticles agglomerate and sinter, decreasing the active surface area. The sintering of the precious metal particles during operation causes a loss of catalytic activity (i.e. deactivation). Exhaust gases exiting from petrol engines change quickly and dramatically, during operation. Temperatures can rise transiently to around 1000°C and the exhaust gas composition itself fluctuates quickly between oxidative and reductive compositions. Therefore, in situ dynamic observation of the sintering and redispersion phenomena of the precious metal in the automotive catalysts is very important indeed.
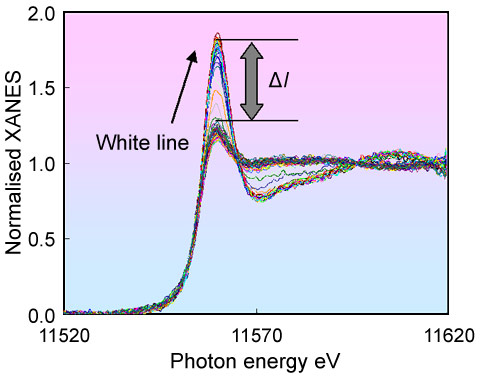
Real-time observation of the sintering/redispersion behaviour of Pt was made possible by the fluorescence yield variant (Turbo-XAS) of energy dispersive X-ray absorption spectroscopy (XAFS) developed by Pascarelli et al. at ID24 [1]. Figure 1 presents a specially designed in situ cell [2] and two detectors for incoming and fluorescence X-rays. The fluorescence yield Turbo-XAS experiments led to Figure 2, where reasonable quality spectra for 2 wt% Pt/Ce-Zr-Y mixed oxide (referred to as CZY) catalysts under in situ conditions could be collected. In our previous study, transmission data for Pt/CZY was very noisy, making quantitative analysis difficult [3], because the combination of low levels of Pt in the catalysts with high levels of heavy, absorbing elements such as Ce and Zr severely compromises the conventional, transmission-based experiment.
 |
|
Figure 1. Catalyst sample heating in an in situ cell for time-resolved XAFS [2] using the fluorescence yield variant (Turbo-XAS). |
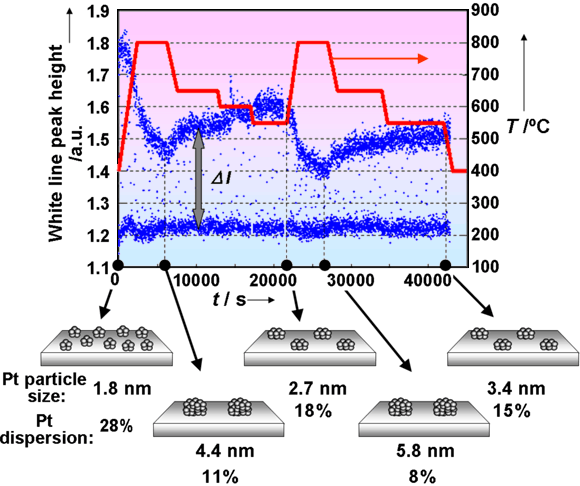
Figure 3 shows the variation of the white line peak height of the normalised Pt LIII edge XANES for the fresh Pt/CZY catalysts under cyclical oxidising/reducing condition at 400 ~ 800°C. ΔI in Figure 3 denotes the difference between the white line peak height of the oxidised and reduced samples. From a previous experiment [3], we found that the ΔI increased with decreasing particle size of Pt. Using the correlation between the Pt particle size and ΔI, we observed that the Pt particle size changed according to the temperature. It is interesting to note that the sintering/redispersion phenomena can be reversibly controlled using the sample temperature. In contrast, only facile sintering of Pt particles is observed in the Pt/Al2O3 case, and Pt redispersion is not achieved at any temperature. As such, this kind of Pt redispersion on CZY is reasonably attributed to the strong Pt-ceria support interaction [4].
Following further kinetic measurements using XAS and in situ transmission electron microscopy, an atomic migration mechanism was proposed to account for the observed redispersion through the trapping of atomic Pt species at sites on the Ce support that exhibit a strong Pt-oxide-support interaction.
Thus the thermo-kinetic character of this process was revealed by the application of time-resolved spectroscopy and microscopy. The results of the study have potential for incorporation into "on board" methodology aimed at extending car catalyst lifetime by curtailing or reversing the effects of metal sintering during operation.
References
[1] S. Pascarelli, T. Neisius, S. De Panfilis, J. Synchrotron Rad. 6, 1044 (1999).
[2] G. Guilera, B. Gorges, S. Pascarelli, H. Vitoux, C. Prestipino, Y. Nagai, N. Hara, submitted to J. Synchrotron Rad.
[3] Y. Nagai, N. Takagi, Y. Ikeda, K. Dohmae, T. Tanabe, G. Guilera, S. Pascarelli, M.A. Newton, H. Shinjoh, S. Matsumoto, AIP conference proceedings. 882, 594 (2007).
[4] Y. Nagai, T. Hirabayashi, K Dohmae, N. Takagi, T. Minami, H. Shinjoh, S. Matsumoto, J. Catal.. 242, 103 (2006).
Principal publication and authors
Y. Nagai (a), N. Hara (b), K. Dohmae (a), Y. Ikeda (b, g), N. Takagi (c, g), T. Tanabe (a), G. Guilera (d, h), S. Pascarelli (d), M.A. Newton (d), O. Kuno (e, i), H. Jiang (e), H. Shinjoh (a), S. Matsumoto (f), In Situ Redispersion of Platinum Autoexhaust Catalysts: An On-Line Approach to Increasing Catalyst Lifetimes?, Angewandte Chemie 47, 9303 (2008).
(a) TOYOTA Central R&D Labs., Inc., Aichi (Japan)
(b) TOYOTA Motor Europe Technical Centre, Zaventem (Belgium)
(c) TOYOTA Motor Corporation Higashi-Fuji Technical Center, Shizuoka (Japan)
(d) ID24, ESRF
(e) TOYOTA Motor Engineering & Manufacturing North America, Inc., Michigan (USA)
(f) TOYOTA Motor Corporation, Toyota (Japan)
(g) Current Address: (f)
(h) Current Address: ALBA-CELLS, Barcelona (Spain)
(i) Current Address: (c)
Acknowledgements
We would like to acknowledge the important contribution of B. Gorges from the ESRF Sample Environment Lab to the design and manufacture of the in situ cell and for the continued advice and assistance during the experiments.