- Home
- News
- General News
- X-rays reveal uptake...
X-rays reveal uptake of nanoparticles by soya bean crops
04-02-2013
Metals contained in nanoparticles can enter into the food chain. Scientists have, for the first time, traced the nanoparticles taken up from the soil by crop plants and analysed the chemical states of their metallic elements. Zinc was shown to dissolve and accumulate throughout the plants, whereas the element cerium did not dissolve into plant tissue. The results contribute to the controversial debate on plant toxicity of nanoparticles and whether engineered nanoparticles can enter into the food chain. The study was published on 6 February 2013 in the journal ACS Nano.
The international research team was led by Jorge Gardea-Torresdey from the University of Texas in El Paso and also comprised scientists from the University of California in Santa Barbara, the SLAC National Accelerator Laboratory in Stanford (California), and the European Synchrotron Radiation Facility in Grenoble (France).
Nanoparticles are present everywhere, for example in the fine dust of wood fires. Even a simple chemical compound behaves differently as a nanoparticle, mostly due to the increased specific surface area and reactivity. These appealing properties are why so-called Engineered Nanoparticles (ENPs) are now widely used in industrial processing and consumer goods. At the same time, their high reactivity has raised concerns about their fate, transport and toxicity in the environment. "A growing number of products containing ENPs are in the market and eventually they will get into the soil, water and air. This is why it is very important to study the interactions of crops with nanoparticles, as their possible translocation into the food chain starts here." says Jorge Gardea-Torresdey, a Professor and Chair of the Department of Chemistry at the University of Texas at El Paso.
The scientists focused on soya bean plants (glycine max), the fifth largest crop in global agricultural production, and the second in the U.S. The soil in which the plants were grown was mixed with zinc oxide (ZnO) and cerium dioxide (CeO2, nanoceria) nanoparticles, which are among the most highly used in industry. ZnO is widely used in sunscreen products, as gas sensors, antibacterial agents, optical and electrical devices, and as pigments. Nanoceria is an excellent catalyst for internal combustion and oil cracking processes and is also used in gas sensors, sunscreen products and cosmetic creams.
After the soya bean plants had been grown to maturity in greenhouses, the distribution of zinc and cerium throughout the plants was studied. The use of microscopic synchrotron X-ray beams at the Stanford Synchrotron Radiation Lightsource (SSRL) and at the ESRF, enabled scientists to determine the chemical form of these metals, i.e. whether they were still bound to nanoparticles or had dissolved and bound with plant tissue. "We used X-ray beams 1000 times thinner than a human hair, and the way in which they are absorbed tells us whether, at the microscopic spot they hit, zinc and cerium were present, and whether they formed part of a nanoparticle in the plant or not." says Hiram Castillo, a scientist at the ESRF in Grenoble.
 |
Soya bean plants during their maturation in greenhouse conditions. Credit photo: J.L. Gardea-Torresdey. |
Cerium was shown to be present not only in the nodules close to the soil but had also reached the plant pods. A detailed spectral analysis of the X-ray signals showed that the cerium in the nodules and pods was in the same chemical state as in the nanoparticles. However, part of the cerium had changed its oxidation state from Ce(IV) to Ce(III) which can alter the chemical reactivity of the nanoparticles.
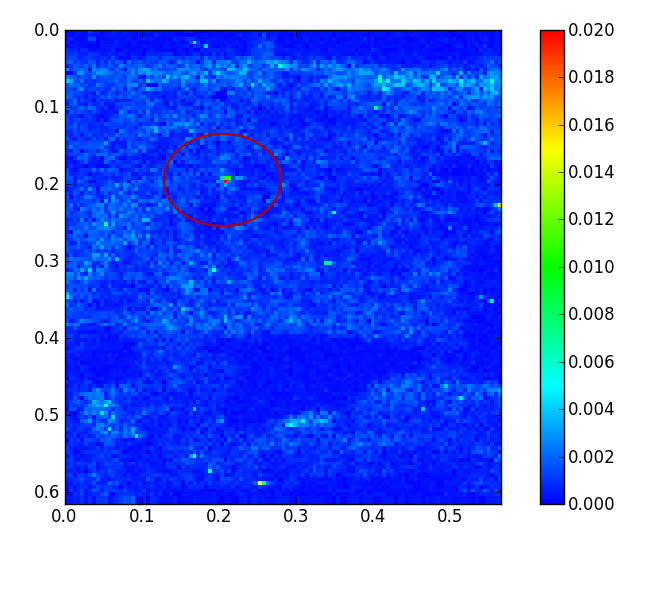 |
Highly sensitive spectral analysis techniques at the ESRF enabled scientists to detect otherwise undetectable nanoparticles of cerium dioxide in the soya beans. Image credit: H. Castillo-Michel (ESRF). |
Zinc was detected in nodules, stems and pods in concentrations higher than in a control group of plants. The spectral analysis did not show the presence of zinc in the plants bound as ZnO nanoparticles which means that the zinc in the nanoparticles had been biotransformed. The spectra suggest that organic acids present in the plants such as citrate, are the probable ligands for the zinc.
"As zinc is present in most plants, it didn’t come as a surprise that zinc from the nanoparticles in the soil can enter into the plant tissue. But plants can also assimilate more dangerous elements like cadmium or arsenic which, when used in nanoparticles, might pose a real threat." says Hiram Castillo. "Our results have also shown that CeO2 nanoparticles can be taken up by food crops when present in the soil. Cerium has no chemical partner in the plant tissue and is not biotransformed in the soya bean but still reaches the food chain and the next soya bean plant generation." adds Jorge Gardea-Torresdey.
"One must keep in mind that once engineered nanoparticles enter the food chain, this is an accumulative process. Tolerable levels today can become dangerous tomorrow. This is why it is important to study not only whether man-made nanoparticles can be taken up from soil but also how they are biotransformed in the plants." concludes Jorge Gardea-Torresdey.
Arturo A. Keller of the University of California in Santa Barbara and Co-Director of the UC Center for the Environmental Implications of Nanotechnology, who was not involved in this research, comments:
“It’s a fascinating paper with some genuine concerns in terms of potential health implications. Whilst we are not able to directly attribute nanoparticle ingestion to any particular disease or symptoms, we know from the latest laboratory studies the potency some have in terms of infiltrating our cells and tissue and causing harm. The fact that these potentially dangerous particles are being taken up by such a common crop suggests a need to review what materials are used in agriculture around the world. In particular, it raises concern over the use of treated waste water to irrigate crops all over the world which may provide a route for these potentially dangerous particles to get into our bodies if the content of the water is not more tightly managed.”
Reference: doi/abs/10.1021/nn305196q
Text by Kirstin Colvin
Top image: Soya bean plants were found to have taken up cerium from the soil. In the plant nodules and pods, the element was found to be in the same chemical state as the nanoparticles present in the soil. Photo credit: Scott Robinson - creativecommons.org



