- Home
- News
- Spotlight on Science
- Structural basis...
Structural basis of membrane protein chaperoning through the mitochondrial intermembrane space
04-12-2018
Mitochondria are involved in cellular metabolism, cancer and apoptosis. 99% of their proteins are produced elsewhere and shuttled to their “work place” in mitochondria. A key step in the import of membrane proteins has now been solved.
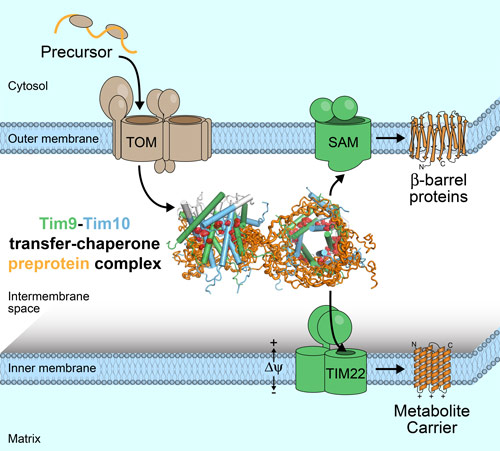
Mitochondria, famous as the “powerhouses” of the eukaryotic cell, are involved in numerous cellular processes including many metabolic pathways, ageing and cell death. The mitochondrial matrix is surrounded by two membranes, and the inner membrane hosts the ATP synthesis machinery. Metabolites and proteins have to be shuttled across these membranes by channels and transporters. The amount of solute that is shuttled across the membranes is enormous; in the case of one, adenosine triphosphate (ATP), 60 kg are transported out of mitochondria every day in a human body. The membrane proteins that perform this transport are themselves not made in mitochondria, but are encoded on nuclear DNA and produced by cytosolic ribosomes, and after their synthesis they have to be brought to their “working place” in one of the two membranes. As all membrane proteins, these polypeptide chains are highly hydrophobic, and in the aqueous compartments through which they have to travel to reach their membrane they would spontaneously aggregate, with dramatic cellular consequences. To avoid such aggregation, eukaryotic cells have developed dedicated “chaperones”, proteins which ensure the safe transport.
This study has focused on the key chaperone in the mitochondrial inter-membrane space, the “translocase of the inner membrane” complex, TIM9·10, which is responsible for chaperoning both inner-membrane and outer-membrane proteins to their respective membranes (Figure 1). The way that these chaperones stabilise membrane proteins in a soluble state has remained elusive, and fundamental points of this process are still not completely understood. Remaining to be discovered is how chaperones hold a membrane protein (in transit to its membrane) tightly, thus avoiding aggregation, while at the same time being sufficiently versatile to do so with many different membrane proteins. Also unknown is how the membrane protein is eventually released once arrived at the insertase, despite the apparently high affinity it has for the chaperone.
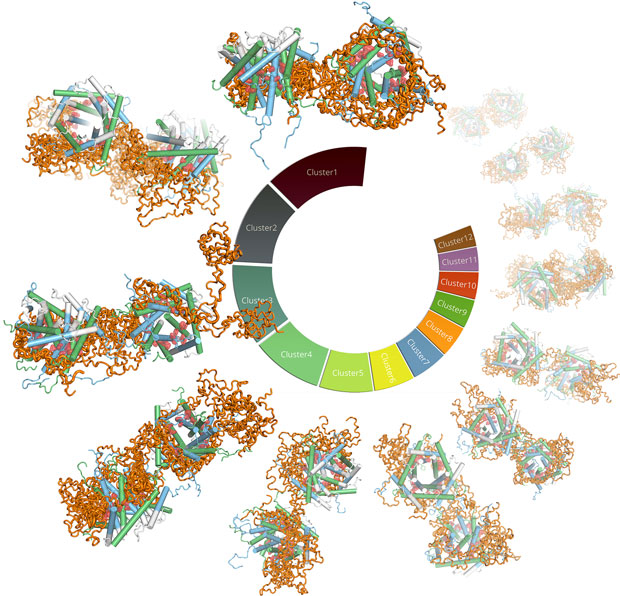
By integrating numerous structural techniques (small-angle X-ray scattering at BM29, NMR), biophysical, biochemical and in-vivo experimental data as well as molecular dynamics simulations, the structure of the TIM9·10 chaperone in complex with a membrane protein was determined at the atomic scale, and thereby the mechanistic details could be revealed (Figure 2). Clamp-like structural elements on the chaperones bind the hydrophobic patches of the membrane protein precursor and hold it in an elongated conformation, but they do so through dynamic exchange of multiple interconverting states. The structural model also allows rationalisation of the mechanistic origin of a genetic disease related to mitochondrial import, the Mohr-Tranebjærg or deafness dystonia syndrome.
 |
|
Movie: The dynamic complex of the TIM9·10 chaperone (green, blue) in complex with GDP/GTP Carrier Ggc1 (orange), as viewed by MD simulations, re-weighted with the experimental data (click to view or download the movie). |
Principal publication and authors
Structural basis of membrane protein chaperoning through the mitochondrial intermembrane Space, K. Weinhäupl (a), C. Lindau (b,c), A. Hessel (a), Y. Wang (d), C. Schütze (b), T. Jores (e), L. Melchionda (b), B. Schönfisch (b), H. Kalbacher (e), B. Bersch (a), D. Rapaport (e), M. Brennich (f), K. Lindorff-Larsen (d), N. Wiedemann (b,g), P. Schanda (a), Cell. 175, 1365–1379 (2018); doi: 10.1016/j.cell.2018.10.039.
(a) Institut de Biologie Structurale (IBS), Univ. Grenoble Alpes, CEA, CNRS, Grenoble (France)
(b) Institute of Biochemistry and Molecular Biology, ZBMZ, Faculty of Medicine, University of Freiburg (Germany)
(c) Faculty of Biology, University of Freiburg (Germany)
(d) Structural Biology and NMR Laboratory, the Linderstrøm-Lang Centre for Protein Science, Department of Biology, University of Copenhagen (Denmark)
(e) Interfaculty Institute of Biochemistry, University of Tübingen (Germany)
(f) European Molecular Biology Laboratory, Grenoble (France)
(g) BIOSS Centre for Biological Signalling Studies, University of Freiburg (Germany)