- Home
- News
- Spotlight on Science
- Asbestos exposed...
Asbestos exposed lung tissue changes revealed by X-ray spectroscopy
10-11-2015
Using a combination of two advanced spectromicroscopy techniques at the ESRF, researchers revealed unique elemental and chemical features that characterise tissue response to asbestos fibres and particulates. The results demonstrate that asbestos and dust in general alter iron homeostasis in the lung, although the effect is more pronounced in the case of asbestos. In addition, these findings are the first evidence for the occurrence of a conformational change in the proteic component of the asbestos coating: this evidence adds new elements for unravelling the link between human exposure to asbestos and long-term pulmonary damage.
A large number of pulmonary diseases are induced by the effects of nano- and micro- particulates in inhalants, in particular cigarette smoke, environmental pollution or as a consequence of occupational exposure. Among environmental and occupational inhalants, asbestos exposure is undoubtedly the most risky condition. Asbestos inhalation causes a chronic inflammation process called asbestosis, which in most unfortunate cases triggers tumoral transformation in the lung and the pleura. Asbestos is considered the most potent carcinogen of mineral origin.
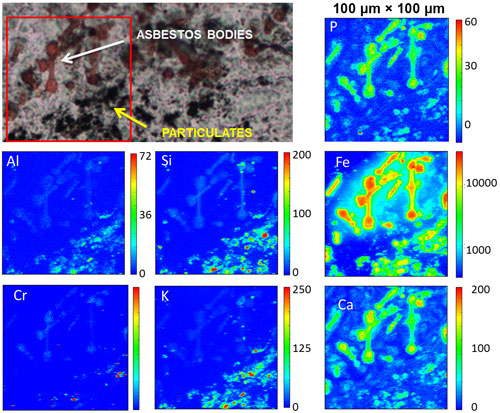
Asbestos exposure is histologically recognised in lung tissues by the presence of the asbestos bodies, which are biopersistent structures formed by the deposition of high quantities of iron containing proteins (mainly ferritin) around the inhaled asbestos fibres (Figure 1). The exact mechanism of formation is still poorly understood, while the ability of asbestos to attract and absorb iron on the surface is recognised as a central process to explain asbestos toxicity. Toxicity and carcinogenicity of asbestos must be connected to the specific chemical composition and physical and surface properties of the material that differentiate asbestos from other inhaled micro and nano pollutants (such as those from urban life).
To unravel the differential body response, a combination of X-ray fluorescence (µXRF) and Fourier Transform infrared (µFTIR) microscopy was used at beamline ID21 to chemically characterise and compare asbestos bodies versus environmental particulates in lung tissues from Italian asbestos exposed patients. Additional µFTIR experiments were carried out at Elettra.
µXRF analyses on the lung tissue samples containing both asbestos bodies and dust particles (Figure 1) allowed the specific elemental composition of these histological structures to be revealed. Dust particles appear to be mainly composed of silicon (Si), aluminum (Al) and potassium (K), with some presence of iron (Fe), and also chromium (Cr). Interestingly, the poor co-localisation of these elements suggests that dust particles of various dimension and composition tend to aggregate/segregate in larger histological formations, as if they were attracted by common surface/chemical properties. In contrast, asbestos bodies are characterised by high levels of iron, calcium (Ca) and phosphorus (P), which cover the thin fibres that are made of silicon. As previously reported [1], calcium and phosphorus are related to the calcification processes that take place in the tissue around the asbestos, as an attempt to isolate the fibres. In contrast, calcifications are not found on dust structures. Another difference in tissue response between asbestos and dust material is demonstrated by the concentration and distribution of iron, which is clearly much higher in the asbestos bodies. The higher level in the surrounding tissue indicates that asbestos alters the iron metabolism in tissue much more than particulates do. The higher presence and mobilisation of iron is in line with the recognised ability of asbestos to produce dangerous oxidative stress conditions in the lung.
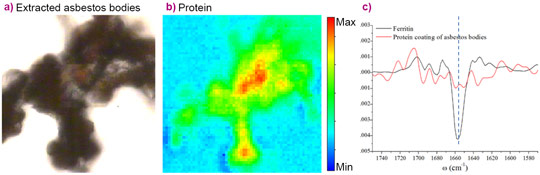
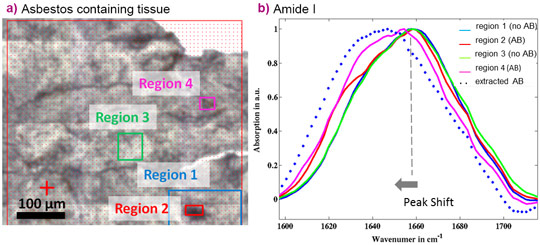
Much more important findings were obtained when µFTIR analyses were used to investigate the peculiar response of tissue to asbestos. The technique revealed that while dust structures in the lung do not attract important proteins to the surface, there is a clear abundance of protein on the asbestos bodies. Amide I and Amide II bands are characteristic for proteins (1720-1490 cm-1), and the secondary structure can be inferred by analysing this infrared region. When investigating the protein characteristics in more depth, it was found that the asbestos coating proteins contain a high level of β-sheet structures (Figure 2). This is intriguing, since the main component of asbestos bodies is ferritin, an α- helix rich protein. The α-helix contribution of ferritin is clearly deducible from the sharpness of the Amide I band centred at 1656 cm-1, while the β-sheet and random-coiled components become more abundant than that of the α–helix on asbestos bodies. The protein conformational characteristics of asbestos bodies have been confirmed by a clear shift in the amide I peak in the collected spectra both for paraffin embedded tissues and freshly extracted asbestos bodies (Figure 3).
It may be speculated that β-structure folding is related to conformational changes occurring over time in the proteins surrounding asbestos fibres and bodies, possibly because of the long-term presence in tissue. Conversely, the protein transition to β-sheets could be a specific chemical mechanism of the asbestos body formation. These findings add new elements to help unravel the link between human exposure to asbestos and long-term pulmonary effects.
Principal publication and authors
Differential protein folding and chemical changes in lung tissues exposed to asbestos or particulates, L. Pascolo (a), V. Borelli (b), V. Canzonieri (c), A. Gianoncelli (d), G. Birarda (e,f), D.E. Bedolla (d,f), M. Salomé (g), L. Vaccari (d), C. Calligaro (h), M. Cotte (g), B. Hesse (g), F. Luisi (i), G. Zabucchi (b), M. Melato (a), C. Rizzardi (j), Scientific Reports 10, 12129 (2015); doi: 10.1038/srep12129.
(a) Institute for Maternal and Child Health, IRCCS Burlo Garofolo, Trieste (Italy)
(b) Department of Life Science, University of Trieste (Italy)
(c) Division of Pathology, CRO Centro di Riferimento Oncologico, National Cancer Institute, IRCCS, Aviano (Italy)
(d) Elettra - Sincrotrone Trieste (Italy)
(e) Lawrence Berkeley National Laboratory, Berkeley (USA)
(f) Physics Department, University of Trieste ( Italy)
(g) ESRF
(h) Servizio Diagnostica Veterinaria, University of Udine (Italy)
(i) Sovrintendenza Medica Regionale, Inail, Trieste (Italy)
(j) Department of Medical, Surgical, and Health Sciences, University of Trieste (Italy)
References
[1] L. Pascolo, A. Gianoncelli, G. Schneider, M. Salomé, M. Schneider, C. Calligaro, M. Kiskinova, M. Melato, C. Rizzardi.. Sci Rep. 3, 1123 ( 2013).
Top image: XRF map of Fe for a 100 micrometre square region of human lung tissue containing asbestos bodies.