- Home
- News
- Spotlight on Science
- Universal solvent...
Universal solvent restructuring induced by colloidal nanoparticles
26-01-2015
Colloidal nanoparticles are widely used in dispersed form for consumer and industrial goods. However, little is known about the chemical environment near the particle surface. Using high-energy X-ray scattering at the ESRF, it has now been shown that nanoparticles subtly influence the local solvent density. Each nanoparticle induces a nanoscopic solvation shell, reaching out as far as 2 nm from the particle surface. This effect is expected to strongly influence the chemistry and rheology of these important materials.
The properties of matter at the nanoscale can differ wildly from that of bulk materials. For example, pronounced increases in chemical reactivity, or changes in optical properties are commonplace. In recent years, nanoparticles with a typical size of 1-100 nm have therefore been used in a wide diversity of applications, for which one common theme can be identified. The nanoparticles are embedded in a matrix that leaves the maximum surface area exposed, and maintains a high degree of dispersion. In many real world applications, for example in sun screen, a liquid or gel carrier is used for this purpose. To prevent agglomeration and so prolong the lifetime of the product, it is important to know how the solvent interacts with the nanoparticles. Likewise, in homogeneous catalysis, where chemical reactions occur at the particle surface, it is important to know whether the local chemical environment is different from that of the bulk solvent.
The reorganisation of solvent molecules around isolated cations has been recognised for many years [1]. Pioneering experiments with synchrotron radiation have shown how bulk planar surfaces can induce surface ordering in fluids [2]. However, no definitive proof previously existed for the formation of a nanoscale solvation shell around colloidal nanoparticles. Nevertheless, such effects have been reported in simulations of solvated nanoparticles [3]. In real systems, this would influence properties including diffusion coefficients and particle growth, which influence the applications described above.
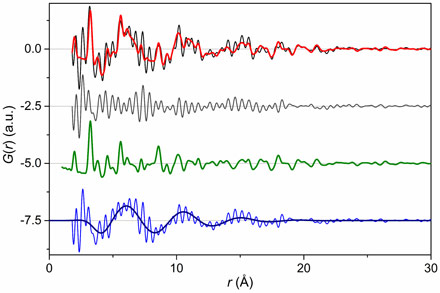
This question has been addressed experimentally using high-energy X-ray scattering at beamline ID15B, in particular using the pair distribution function (PDF) technique. This technique makes use of area detectors to collect scattering data up to a very high resolution in reciprocal space. Fourier transformation then yields the PDF, which is a histogram of all the interatomic distances in the material. Well-defined species, like the ZnO nanoparticles studied here, yield sharp peaks whereas the isotropic density variations found in liquids at medium distances are reflected in a damped oscillation reflecting the average molecular correlations.
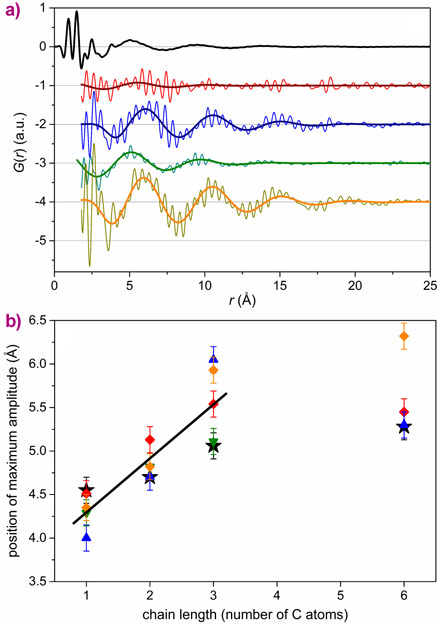
We discovered that the PDF of dry nanoparticles could be well explained by a simple crystal structure model. However, when the nanoparticles were dispersed in a range of solvents, an additional signal appeared. This takes the form of a damped oscillation like that seen in the bulk solvent (Figure 1). However, it is out of phase and cannot be subtracted as a background contribution. After exhaustive experiments, we were able to show that this signal does not depend strongly on particle size, shape, nor the capping agent used. However, a linear trend in the position of the oscillation is found with alkyl chain length in the series methanol-ethanol-propanol (Figure 2). Supplementary measurement on metallic, polar and non-polar oxides showed that the formation of a solvation shell is apparently universal for colloidal nanoparticles.
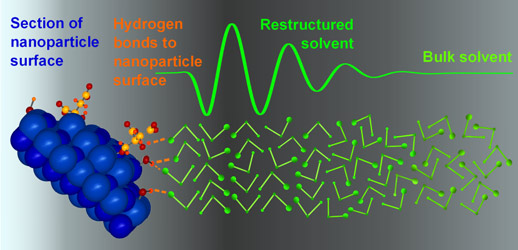
The signal is extremely weak, reflecting both the very low solubility of our nanoparticles (ca. 30-40 millimolar in ethanol), and the proportion of solvent in proximity to the particles. However, we were able to parameterise it using a simple model. The fitting shows that solvent restructuring extends as far as 2 nm into the bulk solvent. Due to the shift of the signal with alkyl chain length, it is likely that the alcohol molecules arrange perpendicular to the surface (Figure 3). Profound changes in the chemical properties of the solvent in this region are thus supported by our analysis.
In summary, we have been able to detect the formation of a nanoscopic solvation shell around colloidal nanoparticles using high energy X-rays. Although the tiny solvent density changes reported here are at the very limit of detectability, when the ESRF phase two lattice upgrade is complete, we will be able to detect changes in nanoparticle size, shape and chemistry, as well as the influence of the solvation shell in real time. This will open the door to a complete understanding of the chemical and rheological properties of solvated colloidal nanoparticles.
Principal publication and authors
Universal solvent restructuring induced by colloidal nanoparticles, M. Zobel (a), R.B. Neder (a), S.A.J. Kimber (b), Science 347, 292-294 (2015).
(a) Department of Physics, Lehrstuhl für Kristallographie und Strukturphysics, Friedrich-Alexander University Erlangen-Nürnberg, Erlangen (Germany)
(b) ESRF
References
[1] D.T. Bowron, S. Diaz-Moreno, J. Phys. Chem. B, 113, 11858 (2009).
[2] O.M. Magnussen, Phys. Rev. Lett., 74, 4444 (1995).
[3] D. Spagnoli, J.P. Allen, S.C. Parker, Langmuir, 27, 1821 (2001).
Top image: Short-range order of solvent molecules at a nanoparticle's surface.