- Home
- Users & Science
- Find a beamline
- X-ray nanoprobe
- ID01 - Microdiffraction imaging
- Science on ID01
- Reactivity and crystallography of a single zeolite crystal
Reactivity and crystallography of a single zeolite crystal
Reactivity and crystallography of a single zeolite crystal revealed by using monochromatic X-rays
Large-scale catalytic processes in the petrochemical industry rely heavily on the catalytic properties of zeolite ZSM-5. Here, we simultaneously investigate structure-reactivity relationship of a single zeolite ZSM-5 crystal using micro-X-ray diffraction (µ-XRD) and µ-X-ray excited optical fluorescence (µ-XEOF) imaging.
Establishing structure-reactivity relationships in heterogeneous catalysis is essential because structural heterogeneities can lead to significant differences in reactivity between catalyst particles. However, to obtain information about the interplay between structure and reactivity at the single particle level is challenging, as it requires a spatial correlation of structural changes of an inorganic material with the chemical reactivity of an organic phase. One approach to achieve such correlation is to collect structure and reactivity information simultaneously within the same measurement. Here we show that it is possible to resolve the complex crystallographic structure of a single zeolite ZSM-5 crystal while simultaneously following the reactivity of organic molecules at catalytically active sites using a monochromatic X-ray beam.
Zeolite ZSM-5 crystals represent a prime example of crystallographic and compositional complexity within a single catalyst particle [1]. A 3-D porous network of Si-O and Al-O tetrahedra ultimately determines the orientation of zeolite pore channels and catalytic properties of the zeolite (i.e., the arrangement of Al atoms in the zeolite framework acting as acidic sites). To study the chemistry of zeolite ZSM-5, large zeolite crystals (100×20×20 µm3) are often used as model systems. These crystals consist of at least six subunits with so-called 90° intergrown crystalline domains. Furthermore, the crystals show pronounced Al zoning, i.e. significantly higher concentration of Al in the 2 µm thick outer rim [2]. As a result of this structural anisotropy, crystalline domains within one zeolite ZSM-5 crystal can be unequally affected by post-treatment methods, such as steaming at elevating temperatures, resulting in distinctly different crystallinity and reactivity.
To study the structure-reactivity relationship of a single zeolite ZSM-5 crystal, we employed a novel characterization approach based on simultaneous micro-X-ray diffraction (µ-XRD) and µ-X-ray excited optical fluorescence (µ-XEOF) imaging (Figure 1). An 8.5 keV X-ray beam was used to acquire both diffraction (XRD) and spectral (XEOF) information, achieving a lateral resolution of ~ 500 nm. With this approach, the local zeolite crystallinity, as measured by µ-XRD, is correlated with the local presence of Brønsted acidity, as measured by µ-XEOF. The latter is based on the use of an X-ray excited photoemission of visible light from the fluorescent products formed upon reaction of 4-methoxystyrene probe molecules with acid sites of zeolite ZSM-5 (Figure 1). The linear products (detected at 610 nm in the XEOF spectrum) form selectively in the micropores of zeolite ZSM-5, whereas bulkier cyclic species (530 nm) readily form at acid sites near crystalline defects, which provide the pore space necessary to accommodate larger products (Figure 1).
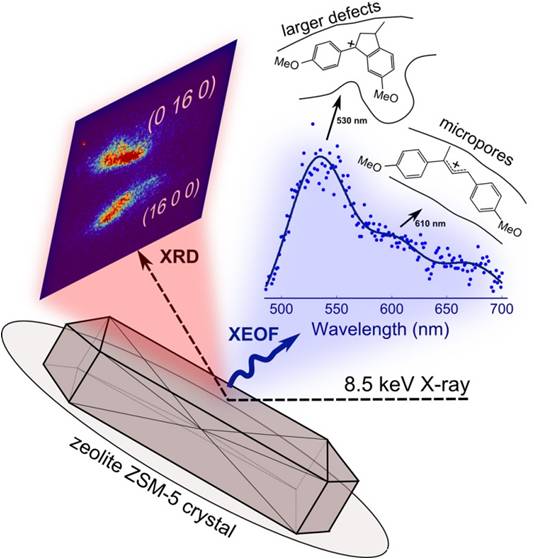
Figure 1. Schematic of the combined µ-XRD/ µ-XEOF imaging approach. An 8.5 keV X-ray is focused on a zeolite ZSM-5 crystal. The (16 0 0) and (0 16 0) Bragg reflections are detected with a 2-D X-ray detector, whereas the XEOF spectrum is collected with a highly-sensitive UV-Vis spectrometer.
We showcased the µ-XRD/µ-XEOF approach on an anomalously intergrown, steamed zeolite ZSM-5 crystal, which contains large structural heterogeneities. As determined by PCA (principal component analysis) and subsequent clustering, the µ-XRD data reveals five distinct intracrystalline domains with different crystallographic orientations and concentration of framework Al (Figure 2a). Consequently, the overlaid µ-XEOF map shows large differences in reactivity (Figure 2b). Several conclusions can be drawn based on the cluster maps. Crystallographic phases within one zeolite crystal are unevenly affected by steaming and show different catalytic activity (Figure 2c,d); this is a result of the described structural anisotropy. The Al-rich outer crystalline domains appeared highly prone to dealumination. In these regions, low to moderate reactivity of the probe molecules was observed, while the inner zeolite domains that were moderately steamed showed significantly higher probe reactivity.
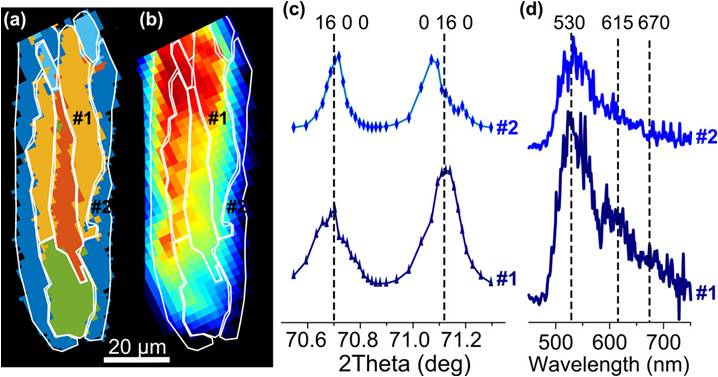
Figure 2. a) PCA cluster map of different crystalline domains within a single zeolite ZSM-5 crystal. b) Corresponding XEOF total intensity map with the white contour plot showing clusters from (a). c,d) Comparison of c) X-ray diffractograms and d) XEOF spectra for the two regions of interest labelled in (a) and (b).
In summary, a single monochromatic X-ray beam can be used to acquire information in both the X-ray and the visible spectral region, which allowed us to study the impact of the crystalline structure and mesoporous defects on the reactivity of a single zeolite crystal. The developed method has the potential to substantiate, synchronously, with both spatial and time resolution, the structure-reactivity properties of many other important functional materials.
Principal publication and authors
Z. Ristanović (a), J.P. Hofmann (a,b), M.-I. Richard (c,d), T. Jiang (a), G.A. Chahine (c), T.U. Schülli (c), F. Meirer (a), and B.M. Weckhuysen (a), Angew. Chem. Int. Ed. (2016)
(a) Inorganic Chemistry and Catalysis, Utrecht University (The Netherlands)
(b) Eindhoven University of Technology (The Netherlands)
(c) ESRF - The European Synchrotron
(d) Aix Marseille Université, CNRS, Université de Toulon (France)
References
[1] L. Karwacki et. al., Nat. Mater. 8, 959 (2009)
[2] Z. Ristanovic et al., Angew. Chem. Int. Ed. 52, 13382 (2013)



