- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Structural biology
- Structure of the ligand-binding domain of the kainate receptor GluK4
Structure of the ligand-binding domain of the kainate receptor GluK4
Ionotropic glutamate receptors play a key role for fast neurotransmission in the central nervous system and have been linked to several neurological diseases [1]. One subfamily of ionotropic glutamate receptors is the kainate receptors that are grouped into low-affinity (GluK1-3) and high-affinity (GluK4-5) receptors. Until recently, no structures were available of the ligand-binding domain of high-affinity receptor subunits. Here, the X-ray structure of the ligand-binding domain of GluK4 with kainite has been solved.
This study provides an example of the strength of combining high-throughput crystallisation and data collection when only small amounts of protein are available and crystal growth is not straightforward. A soluble construct of the ligand-binding domain of GluK4 (GluK4-LBD; Uniprot entry Q01812, residues 415-527 and 652-790 connected via a three residues (KGT) linker) was expressed in insect cells (Hi-5). The KGT linker keeps the parts together that in the full-length receptor are connected to the transmembrane region. A strepII affinity tag attached to the N-terminus of GluK4-LBD was inserted to facilitate purification. Crystallisation screening of GluK4-LBD with kainate was performed at the EMBL HTX Lab, Grenoble, in sitting-drop vapour diffusion experiments at 4ºC. Only small crystal clusters could be obtained and the observed X-ray diffraction patterns obtained at beamline ID30-A1 (Massif-1) were clearly a result of multi-lattice diffraction from the crystal cluster. However, we managed to process the data to 2.05 Å resolution using Xia2 (version 3.6.3) and XDS. GluK4-LBD crystallised as a monomer in space group C2221 and the structure was solved by molecular replacement and refined to R/Rfree values of 19.1%/26.2%.
Thermal stability of the GluK4-LBD in the presence of kainate and (S)-glutamate, respectively, was analysed using a differential scanning fluorimetry (DSF) assay at the HTX Lab according to a published protocol. Melting temperatures (Tm) of 39ºC with (S)-glutamate and 53ºC with kainate were seen. Compared to a model of GluK4-LBD with bound glutamate the crystal structure of kainate in complex with GluK4-LBD shows additional π-stacking and hydrophobic interactions. As hydrophobic forces are known to be strongly correlated with temperature and increase at higher temperatures, these additional interactions explain the favourable Tm in the presence of kainate compared to glutamate. The larger thermal stability of GluK4-LBD with kainate compared to glutamate is consistent with a better binding affinity of kainate (Kd of 1.9 nM) than glutamate (Kd of 25 nM).
 |
|
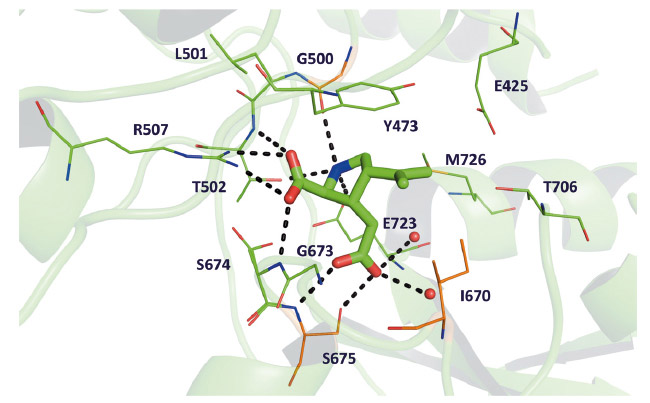
Fig. 73: The binding site of GluK4 comprises 13 residues located within 4 Å of kainate. The three residues not conserved between GluK4 and GluA2 (see main text) are coloured orange. Hydrogen bonding interactions between kainate and GluK4 residues and water molecules (red spheres) are shown as black dashed lines. |
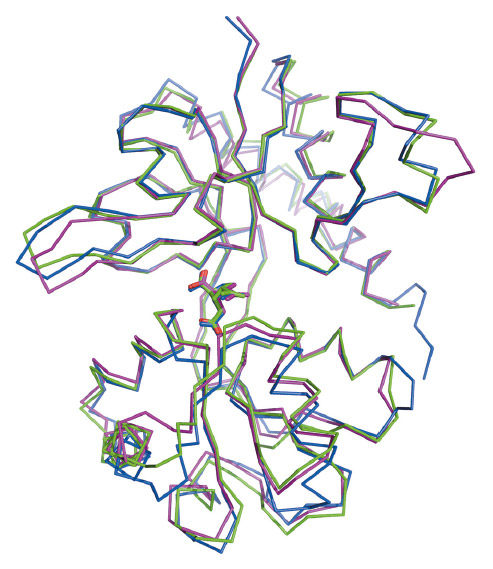
The amino-acid sequence of GluK4-LBD is most similar to those of the other kainate receptors. However, the binding site residues in GluK4 were found to be most similar to another subfamily of the ionotropic glutamate receptors, the AMPA receptor subfamily (Figure 73). Within 4 Å of kainate, only three out of 11 residues differ between GluK4 and the AMPA receptor subunit GluA2: Gly500 (Pro499 in GluA2), Ile670 (Leu671 in GluA2), and Ser675 (Thr676 in GluA2). For comparison, five binding-site residues differ between GluK4 and the low-affinity kainate receptor subunit GluK1. Characteristic features of the GluA2 binding site are conserved in GluK4. These include the presence of Met729 that has been shown to undergo major conformational changes dependent on the actual ligand bound as well as the so-called "interdomain lock" residues Glu423 in lobe D1 and Thr707 in lobe D2 [2]. Thus, the high-affinity kainate receptor subunit GluK4 is different from the low-affinity kainate receptor subunits GluK1-3, which contain a serine (GluK1) or threonine (GluK2-3) at the position corresponding to Met726 in GluK4 and a serine (GluK1) or asparagine (GluK2-3) at the position of Thr706 in GluK4. On the other hand, the degree of closure of the ligand-binding domain around kainate (Figure 74) and interlobe contacts induced by kainate were similar to those in GluK1. The high-affinity kainate receptor subunits are believed to primarily play a role in modifying functional properties of the receptor as they need to combine with one of the low-affinity kainate receptors to assemble, traffic to the membrane surface, and form a functional ion channel. The unique characteristics of the ligand-binding domain of GluK4 support such a functional role.
 |
|
Fig. 74: Overlay of the kainate bound structures of GluK4-LBD (green), GluK1-LBD (magenta) and GluA2-LBD (blue). |
Principal publication and authors
The structure of a high-affinity kainate receptor: GluK4 ligand-binding domain crystallized with kainate, O. Kristensen, L.B. Kristensen, S. Møllerud, K. Frydenvang, D.S. Pickering and J.S. Kastrup, Structure 24, 1582-1589 (2016); doi: 10.1016/j.str.2016.06.019.
Department of Drug Design and Pharmacology, Faculty of Health and Medical Sciences, University of Copenhagen (Denmark)
References
[1] S.F. Traynelis et al., Pharmacol Rev. 62, 405-496 (2016).
[2] J. Pøhlsgaard et al., Neuropharmacology 60, 135-150 (2011).



