- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- X-ray imaging and optics
- Brain tumour and normal vessel responses to synchrotron microbeam radiation therapy
Brain tumour and normal vessel responses to synchrotron microbeam radiation therapy
Microbeam radiation therapy (MRT) is a pre-clinical form of radiosurgery initially dedicated to the treatment of brain tumours [1]. MRT requires very intense X-rays produced by a third generation synchrotron. The treatment is based on spatial fractionation of the radiation dose instead of conventional clinically-used temporal fractionation. Tumours are irradiated with very high doses (~150-625 Gy) by an array of parallel microbeams (~25-50 µm width), 100 to 400 µm spaced on-centre.
Previous preclinical studies on rodents have shown that normal tissue presents a particularly high tolerance to radiation delivered via microbeams and MRT has been proven to have palliative and even curative effects on cerebral tumours implanted in rat brain. These studies showed that MRT yields a better therapeutic index than conventional, non-spatially fractionated, radiotherapy. This has been largely attributed to a stronger effect of microbeam irradiation on tumoural vessels than on normal vessels. It has been hypothesised that MRT-damaged microsegments in poorly differentiated neovasculature of brain tumours may not be efficiently repaired, in marked contrast to the rapid repair of radiation-damaged normal vessels [2,3]. This tumour vessel necrosis would lead to tumour asphyxia and lesion cure.
In this work, we characterised by MRI and histology the acute response of a 9L tumour implanted in the mouse brain (D0) and its vasculature to crossfired MRT performed on day 13 (Figure 133).
 |
|
Fig. 133: a) Schematic representation of the irradiation geometry. Each microbeam array contains 28 microbeams (25 µm width, 211 µm on-centre distance, in-beam dose: 500 Gy). The two orthogonal irradiations produce a composite irradiation volume in the brain of 6 x 6 x 6 mm3 at the tumour site. MR images from one mouse obtained 21 days after tumour implantation: b) T2-weighted image and c) related apparent diffusion coefficient, d) blood volume, and e) vessel size index maps. The tumour, readily visible in the left hemisphere, presents increased ADC, BV and VSI. |
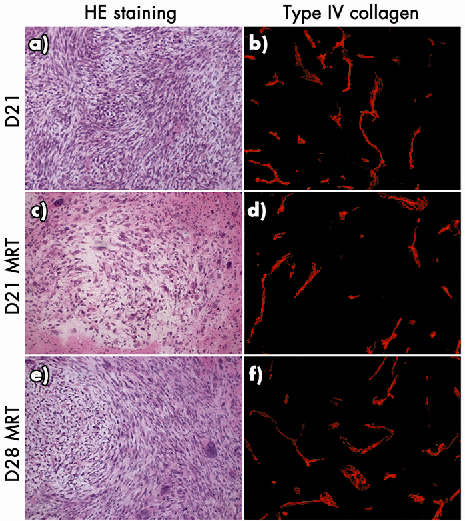
The mean survival time of 9L tumour bearing mice increased significantly for MRT-treated as opposed to untreated groups (x1.34, p<0.0001). A significant increase of apparent diffusion coefficient (ADC) was observed 24 hours after MRT in irradiated tumours versus non-irradiated ones. ADC reflects a radiation-induced increase in tumour vessel permeability which could be exploited to specifically deliver chemo-agents to brain tumours via the circulatory system. Indeed, no change in vessel permeability was observed in the contralateral, unidirectionally irradiated, hemisphere. In the untreated group, both tumour size and vessel size index (VSI, i.e., mean vessel diameter) increased significantly (from 7.6 ± 2.2 to 19.2 ± 4.0 mm2 and +21%, respectively) between the fourteenth and the twenty-first day after tumour cell inoculation. During the same period, in the MRT-treated group, no difference in tumour size was observed. Vessel size index measured in MRT-treated group increased significantly (+26%) but only between 21 and 28 days of tumour growth. We did not observe significant difference in blood volume between the MRT-treated and untreated groups. Histological analysis performed at different periods after irradiation revealed that MRT presented an important cytotoxic effect on tumoural cells while sparing unidirectionally irradiated normal brain tissue (Figure 134). However, tumour vessel labelling (Figure 134) and MRI data suggest that MRT does not reduce blood volume in this tumour model (and therefore the blood supply to the tumour remains practically unchanged), the opposite to expectations from Dilmanian et al. [2].
 |
|
Fig. 134: HE staining a), c), e) and type IV collagen immunochemistry b), d), f) of transversal sections of 9L tumour implanted in nude mice. Untreated (a, b) and MRT-treated (c, d) tumour 21 days after tumour inoculation. MRT treated at D28 tumour (e, f). |
To conclude, this study shows that MRT slows the 9L tumour growth in nude mouse brain. MRI and histological results suggest that the increase in survival time may be due to cytoreduction rather than to a short- term effect of ionising radiation on 9L tumour vessels. Tumour vessels were detected and appeared perfused up to 28 days after irradiation. This implies that our MRT parameters should be optimised and that even higher doses could be delivered to increase the damage to tumour vessels. In this study, measured microvascular parameters suggest that the action mechanism of MRT on 9L gliosarcoma tumour, at least under our experimental conditions, do not involve a significant microvascular component and that the cellular processes involved require further investigation.
Authors
R. Serduc (a,b,c), T. Christen (a,b), J. Laissue (d), R. Farion (a,b), A. Bouchet (a,b), B. van der Sanden (a,b), C. Segebarth (a,b), E. Bräuer-Krisch (e), G. Le Duc (e), A. Bravin (e), C. Rémy (a,b) and E.L. Barbier (a,b).
(a) INSERM, U836, Grenoble (France)
(b) Université Joseph Fourier, Grenoble Institut de Neurosciences, Grenoble (France)
(c) Present address: Centre de Recherche Cerveau et Cognition, UMR5549 CNRS-Université Toulouse, and visiting scientist, ESRF.
(d) Institute of Pathology, University of Bern (Switzerland)
(e) ESRF
References
[1] D.N. Slatkin, F.A. Dilmanian, P. Spanne, Method for microbeam radiation therapy, US Patent (1994).
[2] F.A. Dilmanian, T.M. Button, G. Le Duc, Neuro-oncol 4, 26 (2002).
[3] J.A. Laissue, G. Geiser, P.O. Spanne, et al., Int J Cancer 78, 654 (1998).



