- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- X-ray imaging and optics
- Gold nanoparticles functionalised by gadolinium chelates as contrast agent for both X-ray computed tomography and magnetic resonance imaging
Gold nanoparticles functionalised by gadolinium chelates as contrast agent for both X-ray computed tomography and magnetic resonance imaging
There is a considerable need for better contrast agents both for cancer diagnosis and for monitoring treatments. Commercial contrast agents used in X-ray imaging and magnetic resonance imaging (MRI) – two anatomic techniques widely used to detect tumours – are not sensitive enough to be used with small animals. Indeed, the small molecular size of the contrast agents results in their rapid elimination by the kidneys. Moreover, most of them are specific for one imaging technique alone, whereas to use a combination of imaging techniques would be desirable. Numerous nanoparticulate contrast agents have been recently proposed to overcome these limitations. Their longer vascular half-life renders them suitable for long-term studies of biodistribution, and to evaluate the animal models response to a specific treatment. Another great advantage of the nanoparticle contrast agents lies in their ability to display several complementary properties in the same object. Consequently, some multifunctional nanoparticles that can be detected by several in vivo imaging techniques have been described.
We have recently developed a synthesis of gold nanoparticles functionalised by gadolinum chelates (Au@DTDTPA-Gd). These contrast agents are composed of a gold core (mean diameter of ~ 2.5 nm) encapsulated within a multilayered organic shell of ~ 150 gadolinium chelates (DTDTPA-Gd). These paramagnetic gold nanoparticles are able to induce a contrast enhancement in magnetic resonance images in vitro [1] thanks to the presence of the gadolinium (iii) ions inside the organic shell. The aim of the present study was to investigate the in vivo contrast enhancement of Au@DTDTPA-Gd50 nanoparticles (i.e. Au@DTDTPA nanoparticles with ~ 50 Gd3+ ions trapped in the organic shell) for both MRI (gadolinium ions) and X-ray imaging (thanks to the strong X-ray absorption of the gold core).
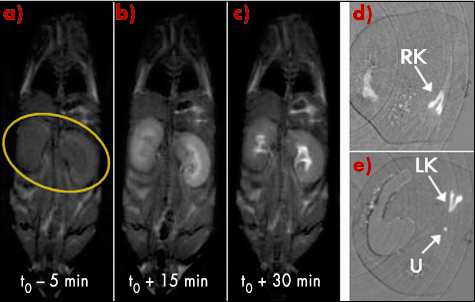
The biodistribution of these nanoparticles was monitored by MRI (at 7T) and X-ray imaging, following their intravenous injection into mice and rats. X-ray imaging was performed at the biomedical beamline (ID17), using two monochromatic X-ray beams bracketing the gold K-edge energy. Both techniques led to the same observations: some areas in the body present an enhanced signal due to the accumulation of paramagnetic gold nanoparticles, this only a few minutes after injection. The contrast enhancement occurs first in the kidneys (Figure 131) and then in the bladder. Thus, the Au@DTDTPA-Gd50 nanoparticles freely circulate within the vascular system without undesirable accumulation in liver and in spleen and are efficiently cleared by renal elimination. This was confirmed by ICP analysis: the gold element was essentially present in the urine and kidneys. Moreover, for both MRI and X-ray in vivo experiments, the contrast induced by these paramagnetic gold nanoparticles allow their monitoring over a long time period compared with molecular contrast agents.
 |
|
Fig. 131: T1-weighted images of a mouse before a) and after b)c) the Au@DTDTPA-Gd50 nanoparticles injection. X-ray images in tomographic mode of a rat at right kidney level d) and left kidney level e). RK and LK for right and left kidneys respectively; U for ureter. |
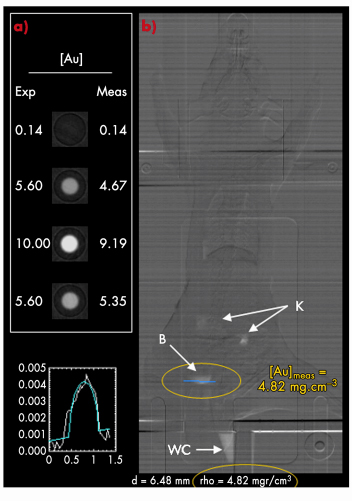
The high photons flux of the biomedical beamline offers two advantages over conventional CT scanners. First, the synchrotron imaging technique requires only low gold concentrations (gold content for X-ray imaging is similar to that used in MRI). Thus, the injection of the contrast agent is less invasive. Another key advantage lies in the quantification of the contrast agent concentration in the tissues. Prior to in vivo imaging studies, in vitro experiments were carried out with aqueous solutions of Au@DTDTPA nanoparticles containing various concentrations of gold (Figure 132a). We can note the concordance between the experimental gold concentration (Exp) and the measurements (Meas) by synchrotron radiation computed tomography (SRCT). The ability to determine quantitatively the gold content in the tissues was used to monitor the biodistribution of paramagnetic gold nanoparticles in vivo and in “real time” (Figure 132b), without sacrifice of the animal. Moreover, another great advantage of the ID17 beamline is its ability to perform radiotherapy [2].
 |
|
Fig. 132: a) Phantom images of samples containing differents gold concentrations (‘Exp’) measured by SRCT (‘Meas’). The last phantom contains Au@DTDTPA-Gd50 nanoparticles. b) In vivo measurement of gold concentration ([Au]meas) in the bladder of a rat by SRCT (blue line). B for bladder; K for kidneys and WC for the tube collecting the urine. |
In conclusion, this study confirms that Au@DTDTPA-Gd50 nanoparticles freely circulate in the vascular system and that they can be used in vivo as a bimodal contrast agent for medical imaging. The MRI contrast enhancement comes from the presence of gadolinium ions in the organic shell of the nanoparticles, whereas the gold core provides strong X-ray absorption. These nanoparticles could therefore be a promising tool for preclinical studies in oncology.
Principal publication and authors
C. Alric (a), J. Taleb (b), G. Le Duc (c), C. Mandon (b), C. Billotey (b), A. Le Meur-Herland (b), T. Brochard (c), F. Vocanson (d), M. Janier (b), P. Perriat (e), S. Roux (a), O. Tillement (a), J. Am. Chem. Soc. 130, 5908 (2008).
(a) LPCML, Villeurbanne (France)
(b) Créatis – LRMN, Villeurbanne (France)
(c) ESRF, Grenoble (France)
(d) Laboratoire Hubert Curien, Saint-Étienne (France)
(e) MATÉIS, Villeurbanne (France)
References
[1] P.-J. Debouttière, S. Roux et al., Adv. Funct. Mater., 16, 2330 (2006).
[2] C. Alric, R. Serduc et al. Gold Bull., 41, 90 (2008).



