- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- X-ray imaging and optics
- Biomedical phase-contrast tomography
Biomedical phase-contrast tomography
X-ray computed tomography (CT) is an invaluable three-dimensional non-destructive imaging method with numerous applications in life and material science. The technique yields excellent results where highly absorbing structures are embedded in a matrix of relatively weakly absorbing material (like bones and tissue in the human body). However, for the differentiation of weakly absorbing soft matter, e.g., pathologies in the soft-tissue structure, the contrast achieved by the currently existing, absorption-based X-ray methods is limited, unless contrast agents are used.
Phase-sensitive X-ray imaging can overcome these limitations and yield good contrast for soft-tissue structures. Phase-contrast imaging relies on refraction, or changes in the angular trajectory of X-rays. Just as light rays bend when they enter water from air, X-rays deflect as they travel through objects of varying densities. We used grating interferometry at ID19 to measure this effect and thus obtain detailed radiographic phase-contrast X-ray images. This approach can be extended to three-dimensional phase-contrast computed tomography (PC-CT) by rotating the specimen around an axis perpendicular to the X-ray beam and recording several hundred differential phase-contrast projections [1,2]. Using specially developed filtered back-projection algorithms, a quantitative three-dimensional volume data set of the specimen can then be reconstructed by computer.
 |
|
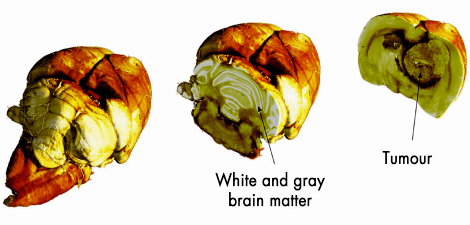
Fig. 129: Post mortem three-dimensional X-ray phase-contrast CT images of a rat brain bearing a 9L gliosarcoma (rat brain tumour), obtained at ID19. The images show three-dimensional renderings of the reconstructed data set. |
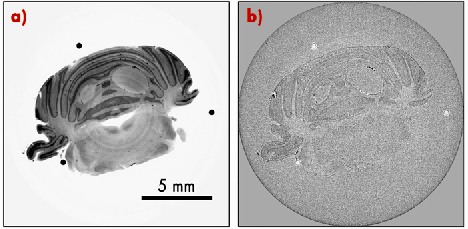
To evaluate the potential impact of this novel grating-based PC-CT method particularly for biomedical imaging applications, we carried out several in-vitro synchrotron experiments on biological specimens. As an example, Figure 129 shows results for a rat brain specimen, fixed in formalin solution. The data reveals two interesting findings. Firstly, there is good contrast between the brain’s white and grey matter. This can be seen in the image of the region containing the rat’s cerebellum (centre). Although such differentiation is rarely possible with absorption-based X-ray CT scans, our method clearly resolves these small density differences between the two brain tissues. Secondly, pathologies such as tumours can be clearly distinguished. The right panel of Figure 129 shows a section through a region in the brain containing a tumour, where we can clearly see the presence of a 9L gliosarcoma (rat brain tumour) in the right caudate nucleus of the brain, even though no contrast agent was used in the experiments. This again confirms the enhanced tissue sensitivity of the method. Figure 130 shows a comparison between the phase-contrast CT results (left panel), and the corresponding tomographic slice obtained by conventional absorption-based CT reconstruction (right panel). Both results were obtained from the same data set, i.e., with the same exposure time.
 |
|
Fig. 130: Reconstructed tomographic slices through the rat’s cerebellum. a) Phase-contrast CT results, b) Conventional absorption CT results. |
With this work we have demonstrated how grating-based phase-contrast CT can yield images of biomedical specimens with unprecedented soft tissue sensitivity thanks to the high spatial coherence of the synchrotron beam at ID19. In particular, we have shown that this X-ray method can be used to discern between subtle details in the tissue structure of animal brains, an application field which until now has almost exclusively been reserved for other techniques such as histology and non-functional imaging techniques. Furthermore, the approach can be combined with local tomography to obtain a detailed view of a region of interest within a large sample. We believe that this method can be applied to a range of biomedical investigations immediately, including studies of the development of certain pathological features associated with neurodegenerative diseases (Alzheimer’s disease), or the complex formation of blood vessels in tumour models. These results are also potentially interesting from a clinical point of view, since a similar approach can be implemented with standard X-ray tube sources [3].
Principal publication and authors
F. Pfeiffer (a,b), C. David (a), O. Bunk (a), T. Donath (a), M. Bech (c), G. Le Duc (d), A. Bravin (d), and P. Cloetens (d), Phys. Rev. Lett. 101, 16810 (2008).
(a) Paul Scherrer Institut, Villigen (Switzerland)
(b) EPFL, Lausanne (Switzerland)
(c) Niels Bohr Institute, Copenhagen (Denmark)
(d) ESRF, Grenoble (France)
References
[1] T. Weitkamp, A. Diaz, C. David, F. Pfeiffer, M. Stampanoni, P. Cloetens, and E. Ziegler, Opt. Express 13, 6296 (2005).
[2] F. Pfeiffer, O. Bunk, C. David, M. Bech, G. Le Duc, A. Bravin, and P. Cloetens, Phys. Med. Biol. 52, 6923 (2007).
[3] F. Pfeiffer, C. Kottler, O. Bunk, and C. David, Phys. Rev. Lett. 98, 108105 (2007).



