- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- Structural Biology
- A Structural Journey into Tubulin Regulation
A Structural Journey into Tubulin Regulation
Microtubules are dynamic components of the cytoskeleton. They rearrange continuously. In particular during cell division, cytoplasmic microtubules depolymerise and reassemble into the mitotic spindle. The heterodimeric ab tubulin proteins are the microtubule building blocks. Microtubule dynamics is regulated by intracellular proteins. Among them, stathmin-like domains (SLDs) of stathmin family proteins sequester two tubulin molecules per SLD in a tight complex (T2-SLD). Stathmin phosphorylation destabilises the complex and releases tubulin. Relatedly, phosphorylation of overexpressed stathmin is required for cells to go through mitosis. Microtubule dynamics is also hindered by exogenous ligands, some of which are anti-cancer drugs.
In the course of our structural studies of tubulin regulation, we have reported a partial structure of a T2-SLD complex [1] at 4 Å resolution, determined by molecular replacement using an electron microscopy model of polymerised tubulin. The limited resolution of the data did not allow us to refine the structure free from model bias. We therefore chose to redetermine the structure from scratch, using experimental phases. Combining a large number of native and derivative data sets measured on ID14-4, careful data collection taking radiation decay into account [2] and improved crystals, we now have a better defined picture of the complex at 3.5 Å resolution. Three main features have emerged from an analysis of this recent structure.
In the complex, the two tubulins are in a curved head-to-tail assembly (Figure 77). In contrast, tubulins are in a straight conformation in microtubules, in which SLD-bound tubulin is not incorporated. Tubulin monomers comprise three domains: N-terminal, intermediate and C-terminal. A comparison of tubulin monomers in the straight and curved structures reveals that they are related by a 10° rotation of their intermediate domains, which drives a translation of the long central helix connecting the N-terminal and intermediate domains. This translation leads to the steric clashes between adjacet tubulin subunits in the microtubules. To avoid these clashes, the neighbouring subunits reorient to yield a curved structure. These rearrangements form the basis of one of the key features of microtubule dynamics, the fast depolymerisation rate. In microtubules, the intermediate domains are locked in place through lateral contacts between neighbouring tubulins. As tubulin molecules start to disassemble, the lateral contacts neighbouring molecules had established with the leaving ones are lost. Their intermediate domains will rotate, lead to curvature and to further loss of longitudinal and lateral contacts. Microtubule destabilisation ensues as well as accelerated depolymerisation.
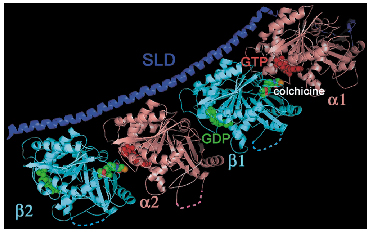 |
Fig. 77: Overview of colchicine-bound T2-SLD. |
The long C-terminal helix of SLD holds the two tubulin heterodimers together while its N-terminal region provides additional stability to the assembly by interacting with the a monomer located at one end of the complex. The N-terminal region forms a ß-hairpin which extends the ß-sheet of the intermediate domain of this ![]() subunit (Figure 78). The N-terminal region will block, together with the curvature of the complex, incorporation of T2-SLD in microtubules. Interestingly, one of the regulatory phosphorylation sites of stathmin is located in the turn of the N-terminal ß-hairpin. An incorporated phosphate group lowers the affinity of stathmin for tubulin, perhaps by destabilising the hairpin.
subunit (Figure 78). The N-terminal region will block, together with the curvature of the complex, incorporation of T2-SLD in microtubules. Interestingly, one of the regulatory phosphorylation sites of stathmin is located in the turn of the N-terminal ß-hairpin. An incorporated phosphate group lowers the affinity of stathmin for tubulin, perhaps by destabilising the hairpin.
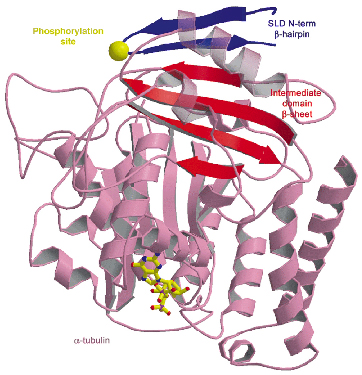 |
Fig. 78: The SLD N-terminal region caps the |
The structure also elucidates features of the mechanism of colchicine interference with microtubules, as the T2-SLD complex was co-crystallised with this small-molecule tubulin ligand. Colchicine is extracted from the crocus flower Colchicum autumnale, whose name derived from Colchis, the living area of the mythical witch Medea. It was widely used to block dividing cells in metaphase well before tubulin was identified as its target. We have localised the colchicine site in the ß tubulin subunit at its interface with the ![]() monomer (Figure 77). Colchicine acts as a wedge that prevents tubulin from adopting a straight conformation and thus inhibits microtubule growth. The structure also accounts for the tubulin stabilising effect of colchicine as it crosslinks the
monomer (Figure 77). Colchicine acts as a wedge that prevents tubulin from adopting a straight conformation and thus inhibits microtubule growth. The structure also accounts for the tubulin stabilising effect of colchicine as it crosslinks the ![]() and ß monomers, rendering tubulin less prone to inactivation. The identification of this binding site and the light we shed on tubulin regulation opens the way for structure-based design of compounds interfering with microtubule dynamics.
and ß monomers, rendering tubulin less prone to inactivation. The identification of this binding site and the light we shed on tubulin regulation opens the way for structure-based design of compounds interfering with microtubule dynamics.
References
[1] B. Gigant, P.A. Curmi, C. Martin-Barbey, E. Charbaut, S. Lachkar, L. Lebeau, S. Siavoshian, A. Sobel, M. Knossow, Cell, 102, 809-816 (2000).
[2] K. Diederichs, S. McSweeney, R.B.G. Ravelli, Acta Cryst. D, 59, 903-909 (2003).
Principal Publication and Authors
R.B.G. Ravelli (a), B. Gigant (b), P.A. Curmi (c), I. Jourdain (c), S. Lachkar (c), A. Sobel (c), M. Knossow (b), Nature, 428, 198-202 (2004).
(a) EMBL, Grenoble ( France)
(b) Laboratoire d'Enzymologie et Biochimie Structurales, CNRS-UPR9063, Gif-sur-Yvette (France)
(c) U440 INSERM/UPMC, Institut du Fer à Moulin, Paris (France).



