- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Structure of materials
- Roughness as a motor for reaction oscillations
Roughness as a motor for reaction oscillations
Oscillatory chemical reactions are often called ‘chemical clocks’ owing to their periodic nature. A well-known system that shows periodic oscillation of reaction rate is the catalytic oxidation of carbon monoxide over platinum and palladium surfaces. We have identified a new mechanism that makes this chemical clock ‘tick’.
A catalyst is a substance that speeds up a chemical reaction without being consumed by the reaction. A notable example is the catalyst in the exhaust of a car, which facilitates the oxidation of poisonous carbon monoxide gas with oxygen to produce the less-harmful carbon dioxide. The rate of conversion of CO to CO2 can spontaneously oscillate in time. A complex interplay of the reactants, CO and O2, modifies the surface structure of the catalyst and thereby its catalytic activity. This causes the rate to oscillate for reactions at low pressures in ultrahigh vacuum systems, as has been beautifully demonstrated by Nobel laureate G. Ertl and co-workers [1]. However, at atmospheric pressures the structure of a catalyst surface is much harder to study because most analytical tools require low-pressure vacuum conditions.
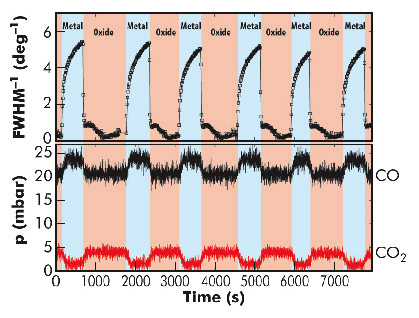
We have developed a novel setup at beamline ID03 that allows the surface structure of a single crystal Pd catalyst inside a flow reactor to be studied during the catalytic reaction by means of surface X-ray diffraction (SXRD) at atmospheric pressure [2]. With these experiments, we show that, during the rate oscillations, the Pd catalyst spontaneously switches between a metallic surface, with a low CO2 production rate, and an oxidised Pd surface, which exhibits a much higher rate, as shown in Figure 40. These observations are in full agreement with earlier studies of the catalytic activity of ultrathin surface oxides.
 |
|
Fig. 40: Top panel: Inverse full width at half maximum (FWHM) of the diffracted intensity at a surface sensitive (anti-Bragg) position as a function of time. The inverse FWHM is proportional to the smoothness of the truncation of the Pd(100) crystal. Bottom panel: partial pressures of CO and CO2 in the reactor. The partial O2 pressure was 500 mbar. The sample temperature was kept constant at 447 K. The colours indicate whether the SXRD intensities identify the Pd(100) surface as a metallic structure (light blue) or as being covered by a thin oxide film (light red). |
Further experiments show that a smooth metal surface oxidises more easily, i.e. under the less-oxidising conditions of a higher CO partial pressure, than a rough metal surface. The authors suggest that this is due to the favourable adsorption of CO at the steps of a rough metallic Pd surface, which thermodynamically stabilises the metal surface. Only when the level of roughness is sufficiently low does the surface becomes oxidised.
 |
|
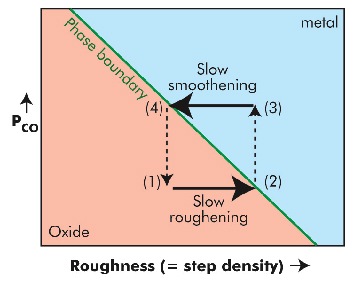
Fig. 41: Metal-oxide stability diagram. Each cycle takes the surface through stages (1) smooth oxide, (2) rough oxide, (3) rough metal, and (4) smooth metal, after which the next cycle starts again at (1). The phase boundary is determined by the roughness and the CO partial pressure. |
The newly discovered mechanism responsible for the oscillatory behaviour, depicted schematically in Figure 41, was found by studying the evolution of the roughness of the metallic and oxidised surface during the oscillations. In the oxide phase of an oscillation cycle, the thin oxide layer becomes gradually roughened by reaction of CO with the oxygen atoms from the oxide layer. This is shown by the broadening of the diffraction peaks. At a certain level of roughness, the oxide layer becomes thermodynamically unstable and disappears, leaving a less reactive metallic surface. This metallic surface becomes smooth again by metal atom diffusion, as shown by the narrowing of the diffraction peak, see Figure 40. The smoothening continues until the roughness is low enough to make the oxide surface thermodynamically stable again, at which point the surface immediately forms a highly-reactive oxide film. This completes the cycle and the process starts all over again from the beginning. Based on these observations, the authors conclude that the evolution of the roughness is responsible for the periodic switching between the metallic and oxidised surface and is at the heart of the mechanism that makes this chemical clock tick.
Principal publication and authors
B.L.M. Hendriksen (a), M.D. Ackermann (a,b), R. van Rijn (a,b), D. Stoltz (a), I. Popa (b), O. Balmes (b), A. Resta (b), D. Wermeille (b), R. Felici (b), S. Ferrer (b,c) and J.W.M. Frenken (a), Nature Chemistry 2, 730–734 (2010).
(a) Kamerlingh Onnes Laboratory, Leiden University (The Netherlands)
(b) ESRF
(c) Present address: CELLS - ALBA, Universita Autònoma de Barcelona (Spain)
References
[1] R. Imbihl and G. Ertl, Chem. Rev. 95, 697-733 (1995).
[2] R. van Rijn, M.D. Ackermann, O. Balmes, T. Dufrane, A. Geluk, H. Gonzalez, H. Isern, E. de Kuyper, L. Petit, V.A. Sole, D. Wermeille, R. Felici and J.W.M. Frenken, Rev. Sci. Instrum. 81, 014101 (2010).



