- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- Surface and interface science
- Tuning intermolecular interaction in long-range ordered sub-monolayer organic films
Tuning intermolecular interaction in long-range ordered sub-monolayer organic films
The future development of organic electronic devices depends on the ability of tailoring the properties of thin-films and their interfaces with (metallic and insulating) substrates. The best results can be expected from well-ordered films with large crystalline grains and “perfect” interfaces. However, their properties are dominantly influenced by the formation of the first molecular layer on the surface representing a template for further growth. The development of the first layer – in turn – depends on the fine balance of molecule-substrate and molecule-molecule interaction. The latter usually is attractive due to van-der-Waals forces and causes formation of islands and small crystalline grains. Here we report on an organic adsorbate exhibiting a different behaviour: Sn-Phthalocyanine molecules show a repulsive intermolecular interaction when they are deposited on a Ag(111) surface in very thin films (coverage below one layer). This ensures a homogeneous filling of the surface terraces and hence the size of structural domains is only limited by the terrace width.
The phase diagram for SnPc/Ag(111) shows three different regions (Figure 85). A disordered overlayer is formed at low coverages and room temperature (RT). Like in a gas, the average intermolecular distance reduces with increasing density. This behaviour is proven by high-resolution low energy electron diffraction experiments (SPA-LEED images are shown for all regions as insets in Figure 85). In the second region at higher coverage, a series of well-ordered phases occurs, the structural parameters of which change continuously with coverage. No discrete phase transitions are found in this regime, but a continuous rotation and deformation of the superstructure unit cell. This effect is caused by a substrate-mediated repulsive inter-molecular interaction between the molecules exceeding van-der-Waals and other attractive forces. The local molecular density is minimised at all coverages. Below 0.92 monolayers (ML), the repulsion can be reversibly switched to attraction by cooling. In this third region an additional site-specific interaction between molecules and substrate over-compensates the repulsion and leads to a rearrangement of the molecules in a commensurate pattern.
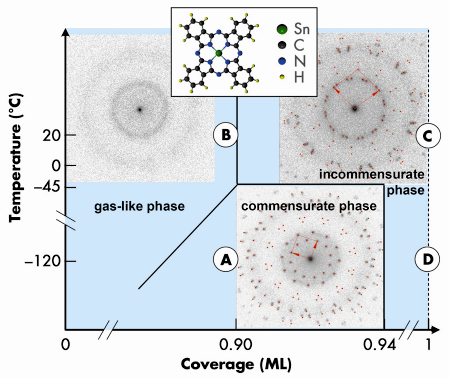 |
|
Fig. 85: Phase diagram of SnPc/Ag(111). Gas-like phases, a region with incommensurate structures, and a commensurate phase are identified. Typical SPA-LEED patterns and a model of the Sn-Phthalocyanine molecule are shown as insets. XSW experiments were performed at the points “A”, “B”, “C” and “D”. |
The key to understanding repulsive interaction is the charge transfer between molecule and substrate. From X-ray standing waves (XSW) measurements performed at beamline ID32, we found convincing indications for a significant overlap of molecular orbitals with the silver electronic states. The adsorption geometry differs for high and low coverages [1]. While in the monolayer-structure, all molecules adsorb with their Sn-atom below the molecular plane (“tin-down” position, Figure 86a), whereas a mixed “tin-up”/“tin-down” structure occurs at lower coverages. The molecule-surface distance is only about 3 Å, significantly below van-der-Waals bonding-distances. This indicates chemisorption and necessitates spatial overlap of molecular orbitals and surface electronic states. An exchange of electronic charge (donation/backdonation) is the consequence. Density functional theory (DFT) calculations and ultraviolet photoelectron spectroscopy (UPS) confirmed this finding.
 |
|
Fig. 86: Structural model and illustration of the donation/backdonation effect. a) Side view of the incommensurate monolayer structure (“C” and “D” in the phase diagram, coverage 1 ML). b) Side and perspective view of the commensurate LT structure (“A”). Molecular orbitals (HOMO-1 and LUMO) are drawn in red and green, respectively. |
The donation/backdonation effect is also illustrated in Figure 86. Electron donation to the substrate just below the molecules (yellow area) displaces Ag electrons to the vicinity (blue). Backdonation to unoccupied orbitals of the molecule cannot completely compensate for this effect. Therefore neighbouring molecules – if they are identically oriented in “tin-down” position and located close to each other as is the case at high coverages (Figure 86a) – compete for the donation/backdonation effect which causes repulsion between the molecules. At lower coverages, the repulsion is reduced by the mixed “tin-up”/“tin-down”-orientation, but at room temperature (disordered, gas-like phase) it still remains dominant. Only upon cooling does the influence of the substrate overcome the repulsion, and an ordered structure is formed (Figure 86b).
Intermolecular repulsion in ordered organic overlayers is reported for the first time. It leads to a novel growth mode based on a homogeneous filling of surface terraces and results in large domain sizes. Just by tuning the temperature, the system can be switched from repulsive to attractive intermolecular interaction and back.
Principal publication and authors
C. Stadler (a), S. Hansen (a), I. Kröger (a), C. Kumpf (a,b) and E. Umbach (a,c), Nature Physics 5, 153 (2009), DOI: 10.1038/nphys1176.
(a) Universität Würzburg, Experimentelle Physik II, Am Hubland, Würzburg (Germany)
(b) present address: Forschungszentrum Jülich GmbH, Jülich (Germany)
(c) present address: Forschungszentrum Karlsruhe GmbH, Karlsruhe (Germany)
References
[1] C. Stadler, S. Hansen, F. Pollinger, C. Kumpf, E. Umbach, T.-L. Lee, and J. Zegenhagen, Phys. Rev. B 74, 035404 (2006).



