- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2008
- Materials science
- Ultrafast X-ray solution scattering reveals a new reaction intermediate in the photolysis of Ru3(CO)12
Ultrafast X-ray solution scattering reveals a new reaction intermediate in the photolysis of Ru3(CO)12
The triangular metal carbonyl cluster Ru3(CO)12 is one of the simplest thermally-stable metal carbonyls. The complex is used in controlled photoactivated synthesis where specific bonds in the complex are broken upon irradiation at different wavelengths. As the mechanism leading to the cleavage of metal-metal bonds is of great theoretical and practical interest, the photolysis of Ru3(CO)12 has been extensively studied by spectroscopy in solid matrices and in solution. Structural characterisation of the intermediates has, however, been very difficult. Recent ultrafast infrared spectroscopy measurements have shown that when solutions of Ru3(CO)12 in non-coordinating solvents like cyclohexane are excited with either an ultraviolet (266 nm) or a visible (400 nm) optical pulse, competing reactions yield two transient intermediates containing bridging carbonyls, Ru3(CO)11(µ-CO) (Intermediate 1) for the metal-metal cleavage reaction channel and Ru3(CO)10(µ-CO) (Intermediate 2) for the CO loss reaction channel, respectively [1]. Infrared spectroscopy specifically monitors the time course of the concentration of these two intermediates via the characteristic absorption bands of their bridging carbonyls. This leaves the possibility that other intermediates could go unnoticed, especially those containing only terminal carbonyls with absorption bands overlapping those of the parent molecule.
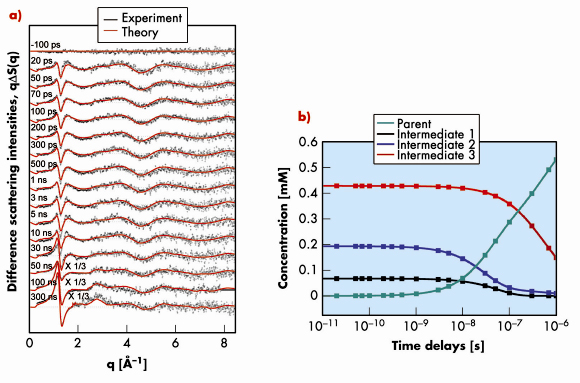
In contrast to infrared spectroscopy, the signal from time-resolved X-ray or electron scattering contains contributions from all interatomic distances in the volume probed by the incident beam. In principle, this makes it possible to detect all transient intermediates [2]. Except for the simplest cases, however, there is no unique solution to recover the three-dimensional structural information from scattering data and this inverse problem can only be solved by modelling based on theoretical calculations. This approach was used to study the photodissociation of Ru3(CO)12 dissolved in cyclohexane in a pump-probe experiment performed on beamline ID09B. Visible (390 nm) laser pulses (2 ps) were used for excitation and 100 picosecond X-ray pulses for probing the transient intermediates. In these experiments the sample flows through a nozzle which produces a thin layer of liquid. The pump-probe sequence is repeated with different time delays between pump and probe at a frequency of 986.3 Hz and the scattered signal is accumulated on a MarCCD detector. The difference X-ray scattering intensities (q![]() S(q,t)) illustrating the structural changes due to the laser excitation are shown in Figure 21a as a function of different time delays.
S(q,t)) illustrating the structural changes due to the laser excitation are shown in Figure 21a as a function of different time delays.
 |
|
Fig. 21: a) Time-resolved difference scattering intensities qΔS(q,t) as a function of time delays after photolysis of Ru3(CO)12 in cyclohexane. The black dots correspond to the experimental data and the red curves to the theoretical least-squares fits. b) Concentration changes of the relevant chemical species during the photoreaction as a function of time. |
Initial attempts at fitting the curves with only the known intermediates 1 and 2 failed to give satisfactory results and suggested the presence of a third intermediate. By modelling the measured difference scattering intensities ![]() S(q,t) using the structures of 16 putative intermediates calculated by density functional theory (DFT), modelled in solution with molecular dynamics (MD) simulations, the concentration of each of the intermediates could be determined (Figure 21b). The results show that three intermediates, Ru3(CO)11(µ-CO), Ru3(CO)10(µ-CO) with bridged CO and Ru3(CO)10 with terminal CO only, are formed at the onset of the reaction from the initial molecule Ru3(CO)12, indicating the rupture of Ru-C and Ru-Ru bonds in Ru3(CO)12 by the optical excitation. The new intermediate Ru3(CO)10 with only terminal CO dominates at all time delays. It recombines non-geminately with one CO ligand to Ru3(CO)10(µ-CO) which eventually decays into the starting molecule Ru3(CO)12 by non-geminate recombination with another CO. Ru3(CO)11(µ-CO) relaxes rapidly to the parent molecule Ru3(CO)12 through geminate recombination.
S(q,t) using the structures of 16 putative intermediates calculated by density functional theory (DFT), modelled in solution with molecular dynamics (MD) simulations, the concentration of each of the intermediates could be determined (Figure 21b). The results show that three intermediates, Ru3(CO)11(µ-CO), Ru3(CO)10(µ-CO) with bridged CO and Ru3(CO)10 with terminal CO only, are formed at the onset of the reaction from the initial molecule Ru3(CO)12, indicating the rupture of Ru-C and Ru-Ru bonds in Ru3(CO)12 by the optical excitation. The new intermediate Ru3(CO)10 with only terminal CO dominates at all time delays. It recombines non-geminately with one CO ligand to Ru3(CO)10(µ-CO) which eventually decays into the starting molecule Ru3(CO)12 by non-geminate recombination with another CO. Ru3(CO)11(µ-CO) relaxes rapidly to the parent molecule Ru3(CO)12 through geminate recombination.
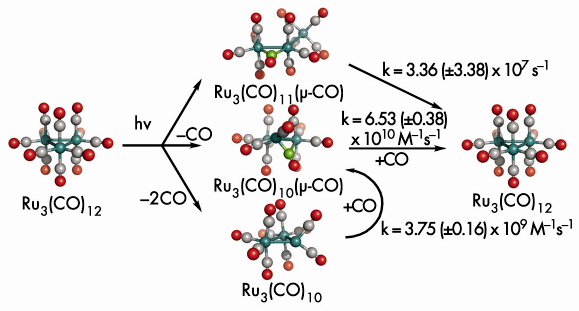
These results strikingly illustrate the complementary nature of ultrafast X-ray scattering and ultrafast spectroscopy. Indeed, a good fit to the experimental X-ray scattering data could only be obtained if intermediate 3, which does not contain any bridging carbonyl and is thus invisible in infrared spectroscopy, is included in the refinement. It would have been very difficult using X-ray scattering alone to establish the existence of the minor intermediate Ru3(CO)11(µ-CO) because its contribution to the overall scattering curve is much smaller than that of Ru3(CO)10. As its existence has been unequivocally established by spectroscopy it is entirely justified to include it in the fit to the X-ray data. Together these techniques clearly indicate the existence of at least three intermediates with very different molecular structures (Figure 22).
 |
|
Fig. 22: Photolysis of Ru3(CO)12 in cyclohexane probed in solution by time-resolved X-ray scattering. Beside the two known intermediates, Ru3(CO)11(µ-CO) and Ru3(CO)10(µ-CO), the hitherto undetected major intermediate Ru3(CO)10 was found. |
Principal publication and authors
Q. Kong (b), J.H. Lee (a), A. Plech (c), M. Wulff (b), H. Ihee (a), M.H.J. Koch (d), Angew. Chem. Int. Ed. 47, 5550 (2008).
(a) Center for Time-Resolved Diffraction, Department of Chemistry, KAIST, Daejeon (Republic of Korea)
(b) ESRF
(c) Fachbereich Physik der Universität Konstanz (Germany)
(d) EMBL, Hamburg Outstation (Germany)
References
[1] E.A. Glascoe, M.F. Kling, J.E. Shanoski and C.B. Harris, Organomet. 25, 775 (2006).
[2] Q.Y. Kong, M. Wulff, J.H. Lee, S. Bratos and H. Ihee, J. Am. Chem. Soc. 129, 13584 (2007).



