- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- Soft Condensed Matter
- Micellar Self-assembly on the Millisecond Timescale
Micellar Self-assembly on the Millisecond Timescale
Amphiphilic molecules possess a unique architecture with a hydrophilic head group and hydrophobic tail(s). When suspended in water, above certain critical concentration these molecules self-assemble into micelles thereby minimising the contact between hydrophobic tail and water. Depending on concentration, temperature and other thermodynamic parameters, a large variety of equilibrium structures such as spherical, cylindrical or disk-like micelles, bicelles, vesicles, and liquid-crystalline phases such as smectic, cubic, hexagonal, and nematic phases can be observed [1]. More recently, these self-assembling properties of amphiphilic molecules have been increasingly exploited in the fabrication of nanostructured materials, e.g. mesoporous systems with well-defined pore sizes. Understanding the dynamical aspects of the micellar self-assembly is often pivotal in their applications because the final state can vary depending on the kinetic pathway. The timescales of the underlying structural organisation may range from a millisecond up to weeks. Small-angle X-ray scattering (SAXS) is a powerful tool that can be used to elucidate the nano-scale structure of these micellar entities. By combining the high brilliance with state-of-the-art detector (RAPID), the very early stages of this self-assembly process, hitherto inaccessible to structural investigations, can be probed.
 |
|
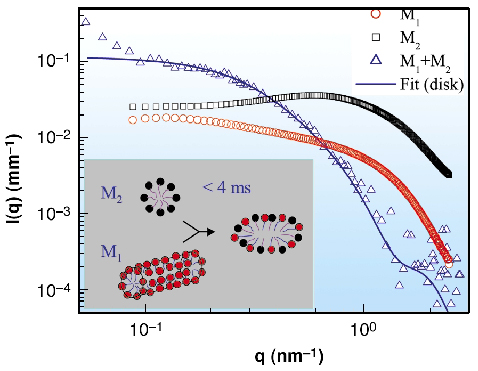
Fig. 47: SAXS intensities for 50 mM solutions of C14H29N(CH3)2O (M1) and C7F15COOLi (M2) and their mixture within the first 5 ms after rapid mixing. The continuous line represents a fit to disk-like mixed micelles. The inset depicts the corresponding structural change. |
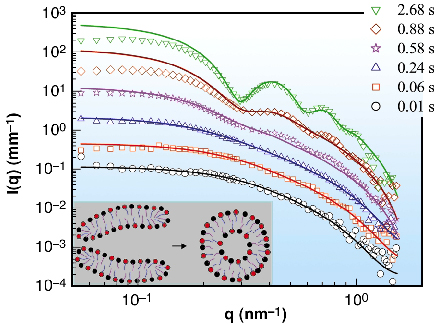
The micelle to vesicle transition is a typical example of micellar self-assembly [2]. For instance, the transition can be induced by rapid mixing (within milliseconds) of equimolar amounts of oppositely charged micelles with a stopped-flow device [3]. Quantitative SAXS data with millisecond range time resolution permits the intermediate states to be accurately modelled. Figure 47 shows the very first step in the transformation of an equimolar mixture of zwitterionic and anionic micelles to vesicles (M1 and M2, respectively). The original micellar solutions contain short rod-like zwitterionic and spherical anionic micelles and they transform to disk-like micelles of twice the size within the mixing time (< 4 ms). Figure 48 further depicts that the radius of these disk-like micelles evolves with time and they close to form unilamellar vesicles within a second. While the details of this transformation are beyond the scope here, it can be mentioned that the driving force is the unstable rim region of the disk-like micelles that generates a line tension which tries to bend the disk to a closed object. This tendency is counterbalanced by the bending energy of the amphiphilic bilayers that tries to keep them flat [2]. From the radius of the initial vesicles (or equivalently the final disk radius) the ratio between bending elastic constants and line tension of the bilayer can be determined.
 |
|
Fig. 48: The time evolution of SAXS intensity illustrating the growth of transient disk-like micelles over the first few hundred milliseconds after mixing and their transformation to unilamellar vesicles within a second. Continuous lines in each of three lower and upper curves represent disk-like micelles and unilamellar vesicles (shell), respectively. For the sake of clarity, the successive scattering curves have been multiplied by a factor |
In summary, the original micelles transform to disk-like micelles in the mixing process. The disks grow in size over a few hundred milliseconds and then close to form vesicles within a second. At longer times the vesicles evolve further and become more polydisperse, but on a much slower timescale. The scattering data demonstrate that the structural changes occur in a very homogeneous and synchronised manner corresponding to a sharp transition. The millisecond time-resolution enables the identification of the different structural intermediates and their dynamics in this multi-step transition. The building blocks of many nano-structured systems are often templated vesicles whose structure is controlled by the dynamics of their formation. In this case for example the monodisperese vesicles are obtained within the first few seconds. Therefore, the micellar structural dynamics can be exploited for the fabrication of advanced complex colloidal materials.
References
[1] W.M. Gelbart, A. Ben-Shaul, and D. Roux, Micelles, Membranes, Microemulsions, and Monolayers, Springer-Verlag, New York (1994).
[2] H.T. Jung, B. Coldren, J.A. Zasadzinski, D.J. Iampietro and E.W. Kaler, PNAS 98, 1353 (2001).
[3] S. Schmölzer, D. Gräbner, M. Gradzielski and T. Narayanan, Phys. Rev. Lett. 88, 258301 (2002).
Principal Publication and Authors
T.M. Weiss (a), T. Narayanan (a), M. Gradzielski (b), C. Wolf (b), P. Panine (a), S. Finet (a) and W.I. Helsby (c), Phys. Rev. Lett. (2005), accepted.
(a) ESRF
(b) Lehrstuhl für Physikalische Chemie I, Universität Bayreuth (Germany)
(c) Daresbury Laboratory, Cheshire (U.K.)



