- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2003
- Macromolecular Crystallography
- Putting Radiation Damage to Good Use
Putting Radiation Damage to Good Use
For a number of years it was believed that the problem of radiation-damage in macromolecular crystallography had largely been overcome by using cryo-cooling techniques. These appeared to give protein crystals an almost endless lifetime on laboratory X-ray sources as well as on beamlines of second-generation synchrotron sources. However, during the design of new beamlines at third-generation synchrotrons, it became clear that even cryo-cooled macromolecular crystals could be destroyed rapidly. Whilst it was realised that it could become difficult to collect highly redundant data sets on these powerful new sources, it was generally believed that radiation-damage would only affect the resolution of the data, and not the structures of macromolecules themselves.
Surprisingly, systematic studies of radiation-damage on cryo-cooled crystals of macromolecules performed at the beamline ID14, showed some highly specific structural changes. These changes include the breakage of disulphide bonds and the decarboxylation of acidic residues [1-3]. Additionally, 'stressed regions' in proteins, such as active site residues or metal ion binding sites, were found to be noticeably more susceptible to the effects of ionising radiation than the rest of the structure. Further studies have confirmed that these effects are general.
Rather than regarding radiation-damage as a problem to be avoided, we have developed a new technique that allows the elucidation of crystal structures of native macromolecules by exploiting the specific structural changes induced upon X-ray exposure. The method has been used to determine the structure of a 23.5 KDa molecular weight protein and that of a synthetic RNA oligonucleotide.
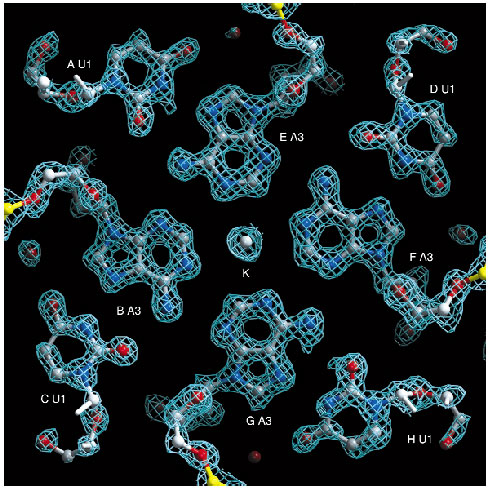
In both cases an initial, complete diffraction data set was collected from the crystals studied. They were then exposed to X-rays for a dose that far exceeded the dose needed for the initial data collection we term this process 'burning' the crystal - after which further complete data sets were collected. Intensity differences between the data sets obtained before and after the 'X-ray burn' could be interpreted using existing software packages and a model of the specific structural differences induced by the X-ray beam was built iteratively. The final model of structural differences was used, in a manner analogous to classic isomorphous replacement techniques, to produce initial estimates of phases and preliminary electron density maps. Powerful automatic model building routines were applied in order to improve these protein phases and to autobuild the entire structure. For the oligonucleotide, the structure could be deduced directly from the spectacularly detailed preliminary electron density maps (Figure 32).
 |
|
Fig. 32: Section of the experimental electron-density map for the oligonucleotide. The map is shown after radiation-damage induced phasing (rip) and solvent flattening. The correlation between this map and the final map after refinement is 0.90. The final model consists of eight copies (labeled A-H) of six nucleotides each. |
We have named this method of structure determination 'radiation-damage induced phasing' (rip). We have derived de novo phases using rip exclusively, but it could also be very useful to exploit the technique in combination with other phasing techniques such as anomalous dispersion. Current developments in crystallographic theory, hardware and software should make the technique more powerful. Programs are being adapted to deal with data collected during the 'burn' of the crystal. These developments make us optimistic that the use of rip, either as primary or secondary source of phase information, will help to render the phase problem a less significant barrier to macromolecular structure determination.
References
[1] W.P. Burmeister, Acta Cryst D56, 328-41 (2000).
[2] R.B.G. Ravelli and S.M. McSweeney, Structure Fold Des 8, 315-28 (2000).
[3] M. Weik, R.B.G. Ravelli, G. Kryger, S.M. McSweeney, M.L. Raves, M. Harel, P. Gros, I. Silman, J. Kroon and J.L. Sussman, Proc Natl Acad Sci USA 97, 623-628 (2000).
Principal Publication and Authors
R.B.G. Ravelli (a), H.K. Leiros (b), B. Pan (c), M. Caffrey (c) and S.M. McSweeney (b), Structure (Camb) 11, 217-24 (2003).
(a) EMBL, Grenoble Outstation (France)
(b) ESRF
(c) The Ohio State University, Columbus (USA)



