- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2003
- High Resolution and Resonance Scattering
- Joint Study of Structure and Dynamics across the First-order Phase Transition in Fluid Phosphorus
Joint Study of Structure and Dynamics across the First-order Phase Transition in Fluid Phosphorus
Liquid-liquid transitions, i.e. first-order transitions between two liquid states characterised by different local structures and thermodynamic properties, are common in multi-component systems and liquid crystals, where composition and molecular orientation are the respective order parameters. Recently, the possible occurrence of liquid-liquid transitions in pure, isotropic liquids has been suggested [1], thus stimulating the development of models and computer simulations, where these transitions are indeed possible. From the experimental side, however, the support for the existence of these transitions is still rare and even controversial, mainly as a consequence of the technical difficulties of the experiments reported so far. However, a remarkably clear example of a pure, isotropic system displaying a liquid-liquid transition has recently been discovered in phosphorus [2]. There, an abrupt, pressure-driven, reversible structural transformation between a low-pressure molecular liquid and a high-pressure polymeric liquid has been reported at a temperature of 1050°C and a pressure of about 1 GPa [2]. Here we further study this interesting case with the aim of characterising the structural and dynamical changes across the transition with diffraction and inelastic X-ray scattering experiments, respectively.
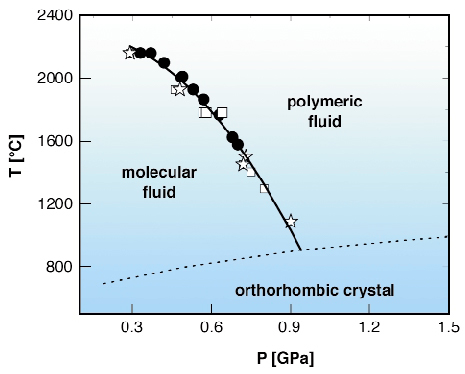
The diffraction experiment has been carried out at beamline ID30 using a Paris-Edinburgh large volume apparatus to reach the extreme thermodynamic conditions of interest here. As the diffraction patterns of the two fluids of phosphorus are clearly distinguishable [2], we could measure the transition line over an extended temperature range up to 2200°C at 0.3 GPa (Figure 6). The addition of the high-pressure high-temperature data to the known phase diagram of phosphorus allows us to conclude that this is a first order transition between a dense molecular fluid and a polymeric fluid. The slope of the transition line is negative in the whole of the explored thermodynamic range, which implies that the entropy of the polymeric fluid is larger than that of the molecular one. Actually, from the shape of the transition line, a thermodynamic characterisation of the transition is obtained in terms of latent heat and internal energy change.
 |
|
Fig. 6: Phase diagram of phosphorus at high temperature and pressure. Stars correspond to coexistence points; solid circles and open squares correspond to data in the polymeric and in the molecular phase, respectively; the full line is the second order polynomial that best represents the transition line; the dotted line is the melting line of orthorhombic phosphorus. |
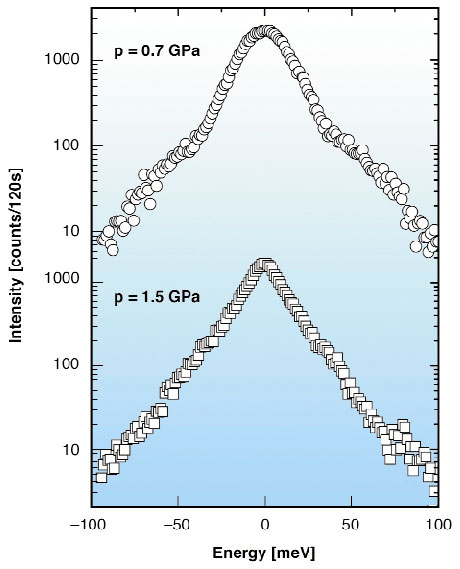
The Inelastic X-ray Scattering (IXS) experiment has been carried out at ID16 using a high-pressure set up similar to that used for the diffraction measurements. IXS spectra of fluid phosphorus were collected on both sides of the first-order transition at T = 1300°C and p = 0.7 GPa in the molecular fluid phase and at T = 1300°C and p = 1.5 GPa in the polymeric fluid one. At each of these thermodynamic points a series of spectra was collected for several q values between 2.0 and 13.0 A1. For q values up to 4.3 A1 the spectral intensities of the two fluids show pronounced differences (Figure 7) which can be interpreted in terms of their different vibrational dynamics. At higher q, however, the spectral intensities of the two fluids become more and more similar, thus suggesting that the polymeric fluid keeps a local structure very similar to that of the molecular one. These results, together with the thermodynamic characterisation of the transition, give us hints on how to better understand the nature of this unusually abrupt polymerisation process in fluid phosphorus.
 |
|
Fig. 7: Inelastic X-ray scattering spectra of the molecular form of fluid phosphorus at T = 1300°C and p = 0.7 GPa (upper spectrum) and of the polymeric form of fluid phosphorus at T = 1300°C and p = 1.5 GPa (lower spectrum). Both spectra have been collected at q = 4.3 A1. |
References
[1] P.H. Poole, F. Sciortino, U. Essmann, and H.E. Stanley, Nature, 360, 324-328 (1992).
[2] Y. Katayama, T. Mizutani, W. Utsumi, O. Shimomura, M. Yamakata, and K.A. Funakoshi, Nature, 403, 170-173 (2000).
Principal Publications and Authors
G. Monaco (a), S. Falconi (b), W. Crichton (a), M. Mezouar (a), Phys. Rev. Lett., 90, 255701 (2003); S. Falconi (b), G. Monaco (a), W. Crichton (a), M. Mezouar (a), R. Verbeni (a), in preparation.
(a) ESRF
(b) Universita' di Roma Tre (Italy)



