- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2002
- Industrial and Applied Research
- Small-angle X-ray Scattering Studies on Concentrated Surfactant Mixtures
Small-angle X-ray Scattering Studies on Concentrated Surfactant Mixtures
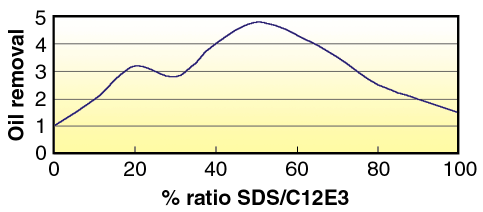
For any company whose business performance is assessed by sales growth, the drive to improve products is paramount to satisfy both the increasingly complex consumer demands and the need to grow market share. However the introduction of a new ingredient to a complex mixture such as a fabric-washing product necessitates a detailed understanding of the interplay between different components. For example the use of two different surfactants leads to a synergistic benefit in "Detergency" as derived by the ability to remove an oil from a substrate (Figure 114), where the mixed surfactant is more efficient at removing oily soil than either of the individual ingredients alone.
 |
|
Fig. 114: Example of synergistic detergency in a mixed anionic nonionic surfactant system. |
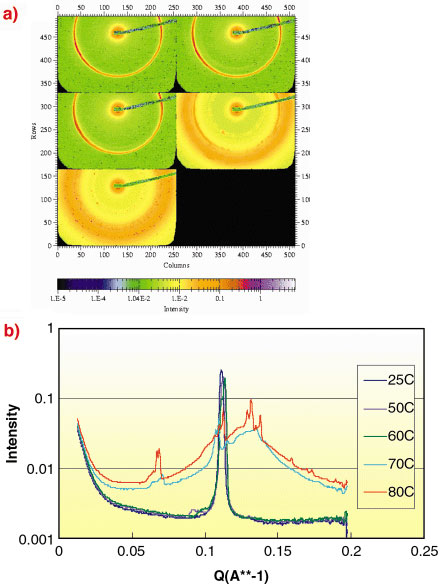
Understanding how the surfactants mix/compete at interfaces is very important and detailed adsorption and adsorbed layer structural studies in the dilute state have been made by combinations of neutron reflectivity and small-angle neutron scattering in order to probe the role of non-ideal mixing in this synergistic effect [1,2]. Ultimately though a company like Unilever must put the ingredients in concentrated form into a product. It is here and also during processing that issues can arise as hot concentrated surfactants exhibit high bulk viscosities via formation of complex mesophases. Data in Figure 115 show how a well-ordered (but low viscosity) lamellar phase can invert to a complex multiphase mixture including a micellar and very viscous cubic phase at high temperature.
 |
|
Fig. 115: a) Typical change in scattering behaviour for concentrated surfactant mixtures with temperature. b) The line plots are radial averages of the 2D images shown in a). Data obtained during commercial beam time on ID2 (June 2002). |
Although these high-temperature data are at a glance quite complicated, the scattering expert is able to resolve features which indicate the presence of different coexisting microstructures. Knowledge of the phase diagram is therefore a prerequisite for processing and this in turn demands accurate measurements at a variety of concentrations, compositions and temperatures. It is here that we value the facilities offered at the ESRF. Specificaly, the beamline ID02 offers the necessary sensitivity/contrasts and q-range, combined with well-controlled sample temperature. When we add source reliability, the sheer speed of data acquisition available on ID02 together with the added benefit of the wide-angle scattering capability, the beamline fulfills and needs. In fact, we have found that we can effectively map out the detail on a complex phase diagram in a single shift of 8 hours.
References
[1] E.J. Staples, L. Thompson, I.M. Tucker, J. Penfold, R.K. Thomas, J.R. Lu, Langmuir 9, 1651, (1993).
[2] E.J. Staples, J. Penfold, I.M. Tucker, J. Phys. Chem. B. 104(3), 606-614, (2000).
Authors
I.M. Tucker, P. Carew, M. Cooke, N. Lewkowicz.
Unilever Research and Development Port Sunlight, Bebington (UK)



