- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2012
- Structure of materials
- Oxygen storage and release at work in catalysis for pollution abatement: illuminating synergies between noble metal and oxide promoter
Oxygen storage and release at work in catalysis for pollution abatement: illuminating synergies between noble metal and oxide promoter
Many modern functional materials are made of multi-phase, nanoscale entities wherein cooperative behaviour between different components is required to obtain optimal performance. Prototypical of such situations are modern three-way catalysts which are utilised in vehicle exhausts to reduce exhaust gas emissions. They convert the three main pollutants in vehicle exhaust, carbon monoxide (CO), unburnt hydrocarbons (HC) and oxides of nitrogen (NOx), to H2O, CO2 and nitrogen. Three-way catalysts comprise a support (Al2O3) upon which both noble metal nanoparticles (Pd, Pt, Rh) and promoting oxides (e.g. CeZrO4) are deposited. Key to the desired catalytic conversions is the structure-reactivity interplay between the promoting oxide and the noble metals, in particular regarding the storage/release of oxygen under process conditions.
We have addressed these interactions at beamline ID15B by combining techniques to achieve bulk, surface and catalytic sensitivity. The techniques used were high-energy X-ray diffraction (HEXRD), infrared spectroscopy (IR) and mass spectrometry (MS). This combination aims to render complete information concerning all solid and gas phases as well as solid-gas interface relevant phenomena. The behaviour of three-way catalysts was thus analysed in dynamic conditions where the gas phase in contact with the catalyst changes from reducing (CO) to oxidising (NO) conditions. The use of HEXRD [1,2] provides kinetic assessment of the above mentioned interactions as well as delineation of how they come together to yield superior catalysts.
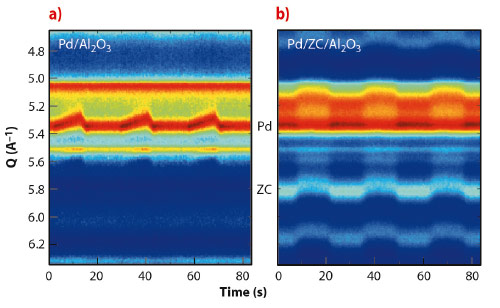
Figure 125 shows time-resolved HEXRD data for two samples with approximately equal Pd particle size in the absence or presence of the CeZrO4 promoter (ZC). The Pd/Al2O3 case (ZC free, Figure 125a) shows the dynamical response of the supported Pd nanoparticles to the change of environment: during CO exposure the Pd lattice parameter expands monotonically (Bragg peaks shift to lower Q).
This behaviour is rapidly reversed under NO resulting in a “sawtooth” variation of the lattice parameter. This is due to the sequential storage and removal of atomic carbon by the Pd nanoparticles [1,2].
The influence of the ZC promoter in Pd chemistry, particularly in its interaction with CO, becomes evident from Figure 125b.The response of the Pd phase to CO/NO gas in the presence of ZC is minimal, but big variations in the ZC lattice parameter as a function of the reducing or oxidising environment can be detected.
 |
|
Fig. 125: Variation in HEXRD (colour map) observed during CO/NO cycling at 673 K over two Pd-containing samples. The experiment shows three cycles comprising a CO/NO treatment of 2 x 13.86 s. |
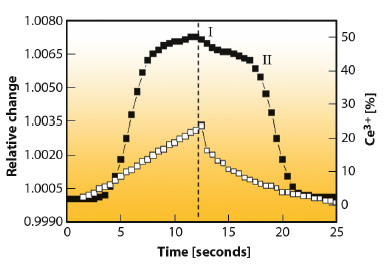
In Figure 126 we present the changes in the ZC lattice parameter and the corresponding redox states, related to Ce3+ formation/annihilation during the same CO/NO dynamical cycle. The Pd-based three-way catalyst system (Pd + ZC + Al2O3) is compared with the support consisting of the ZC promoter deposited onto the alumina carrier. The plot quantifies the oxygen dynamically stored and released and establishes the kinetic dependence of both phenomena. The presence and magnitude of the synergy between Pd and the CeZrO4 are evident. Without noble metal, CeZrO4 donates or stores a limited amount of oxygen. The presence of Pd significantly promotes the process of oxygen transfer in both directions (uptake and release), leading to greatly enhanced catalytic operation.
 |
|
Fig. 126: Relative change of the ZC lattice constant and corresponding Ce3+ content during a CO/NO single cycle at 673 K for the Pd/ZC/Al2O3 catalyst (full symbol) and ZC/Al2O3 support (open symbol). |
From the XRD measurements we also observe precisely when the oxygen donating capacity of the CeZrO4 is exhausted and from MS and IR (not shown) the ramifications this has on the speciation present at the surface of the catalyst. The occurence of oxygen transfer arising from the CeZrO4 radically changes the behaviour of the Pd nanoparticles toward CO and NO molecular and dissociative adsorption, and indeed the formation or not of PdCx phases and or adsorbed CN and NCO molecules during the catalysis. All of these species have fundamental roles to play in the successful removal of pollutant species in the applied process.
Principal publication and authors
M.A. Newton (a), M. Di Michiel (a), A. Kubacka (b), A. Iglesias-Juez (b) and M. Fernández-García (b), Angew. Chem. Int. Ed. 5, 2363-7 (2012).
(a) ESRF
(b) CSIC, Madrid (Spain)
References
[1] M.A. Newton, M. Di Michiel, A. Kubacka and M. Fernández-García, J. Am. Chem. Soc. 132, 4540 (2010).
[2] A. Iglesias-Juez, A. Kubacka, M. Fernández-García, M. Di Michiel and M.A. Newton, J. Am. Chem. Soc. 133, 4481 (2011).



