- Home
- Industry
- Applications and case studies
- Catalysis and Chemistry
Catalysis and Chemistry
The ESRF is helping the chemical industry find more efficient and environmentally compatible processes for the manufacture of materials
Heterogeneous catalysis controls about 90% of the world’s chemical manufacturing processes, yet fundamental chemical and physical mechanisms are often not completely understood and its study remains largely empirical.
Synchrotron techniques at the ESRF offer unique opportunities to understand the behaviour of catalytic systems.
X-ray absorption spectroscopy provides information about the chemical bonds surrounding an absorbing atom, for instance, while qualitative and quantitative measurements during catalysis are possible at millisecond time resolution across all concentration ranges.
• Analyse the shape, size and density of particles including nanoparticles.
• Obtain 3D images of matrices and detect trace elements at high resolution.
• Determine quantitative oxidation states of species at ultra-dilute concentration.
• Study catalytic reactions such as the methanol-to-olefin process in situ and operando at extreme conditions and short timescales.
• Probe reaction intermediates.
• Characterise working batteries and fuel cells.
Company or institute
SINTEF Industry and VITO
Challenge
To characterise a novel 3D-printed metal organic framework (MOF) based material and observe changes in the material during CO2 adsorption.
Sample
The UTSA-16 material was synthesised, formulated as an ink, and then printed in the form of a cylindrical ‘log pile’. This monolithic material was produced from a non-aqueous cellulose-based ink formulation without sintering.
Solution
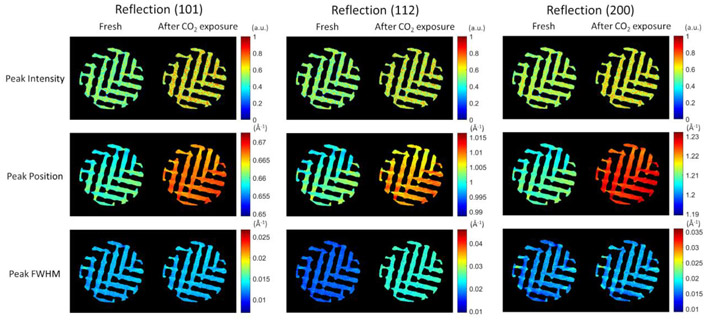
High-energy X-ray diffraction computed tomography (XRD-CT) studies at beamline ID15A can be used to provide both structural and chemical information on a sample within a reactor under operando conditions.
Benefits
In situ high-energy XRD-CT provided insight into the crystal structure and spatial and temporal distribution of active sites within the gas adsorbing 3D-printed material. The study was the first of its kind to explore adsorption in an operating 3D-printed MOF monolith under a CO2 flow. It revealed that the UTSA-16 phase within the 3D-printed monolith changes its structure on CO2 adsorption.
Reference
Multiscale investigation of adsorption properties of novel 3D printed UTSA-16 structures, C.A. Grande, R. Blom, V. Middelkoop, D. Matras, A. Vamvakeros, S.D.M. Jacques, A.M. Beale, M. Di Michiel, K.A. Andreassen, A.M. Bouzga, Chemical Engineering Journal 402, 126166 (2020); DOI: 10.1016/j.cej.2020.126166.
Company or institute
University of Torino in collaboration with Chimet SpA.
Challenge
To resolve the active species on the surface of a platinum catalyst during hydrogenation reactions.
Sample
The sample was an industrial 5 wt% Pt/Al2O3 catalyst of 1.4 nm Pt particle size. The model reaction studied was the hydrogenation of toluene to methylcyclohexane.
Solution
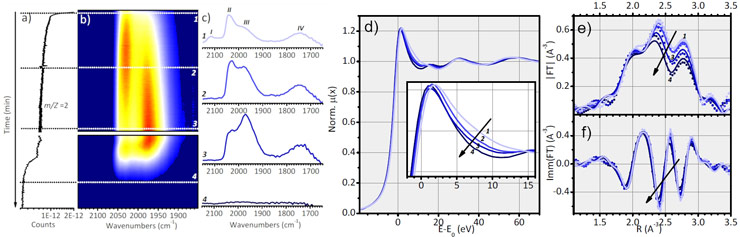
Multiple techniques were necessary to provide a complete picture of the hydrogenated Pt/Al2O3 catalyst. Firstly, inelastic neutron scattering (at ILL, Grenoble) was used to reveal the presence of n-fold coordinated platinum hydrides. FT-IR aided in identifying four linear platinum-hydride species, some of which form at the expense of the n-fold ones when the hydrogen concentration drops. It was confirmed by a combined DRIFTS/XAS/MS experiment at beamline BM23. The XAS part of this experiment (XANES and EXAFS)demonstrated that the Pt nanoparticles undergo a slow and progressive reconstruction upon hydrogenation/dehydrogenation. A second part of the study involved the hydrogenation of toluene, also followed by a combination of DRIFTS/XAS/MS. This demonstrated that the structure of the hydrogenated catalyst remained unchanged, with only surface species participating in the reaction.
Benefits
The combination of techniques confirmed the reconstruction of the catalyst during activation and revealed the platinum-hydride species involved in catalytic hydrogenation and their interplay, which maintains a small proportion of the most active species available as the catalyst for the hydrogenation reaction.
Reference
Dynamics of reactive species and reactant-induced reconstruction of Pt clusters in Pt/Al2O3 catalysts, M. Carosso, E. Vottero, A. Lazzarini, S. Morandi, M. Manzoli, K.A. Lomachenko, M. Jimenez Ruiz, R. Pellegrini, C. Lamberti, A. Piovano, and E. Groppo, ACS Catalysis 9, 7124-7136 (2019); doi: 10.1021/acscatal.9b02079.