- Home
- News
- Spotlight on Science
- Radiation damage...
Radiation damage in serial protein X-ray crystallography
07-07-2020
Sequential serial raster-scanning crystallography with a microfocused synchrotron beam was used to study the effects of radiation damage at room temperature for different doses of radiation in order to determine a dose limit tolerated by the protein crystals under study.
X-ray crystallography is the most prolific method used to determine the structure of biological macromolecules – i.e. proteins, DNA, RNA and complexes thereof. It is limited by the high level of damage inflicted on biological macromolecules by exposure to X-rays. To mitigate damage progression, crystallographic data collection has for the past decades been carried out mostly at cryogenic temperatures, although at the risk of blocking conformational heterogeneity that can be central to biological function. The advent of serial crystallography – whereby each crystal is only exposed once, enabling distribution of the absorbed dose over a myriad of crystals – has now allowed crystallographic experiments at room temperature to become again more frequent, with the promise of permitting time-resolved experiments on a large number of crystalline systems. The pre-requisite is, however, to have determined the maximal X-ray dose that can be safely absorbed by a crystal at room-temperature without compromising the biological information. For the sake of optimising data collection protocols, it is furthermore of interest to know if the time period over which a particular dose is absorbed is important, i.e. whether there is a dose-rate effect or not.
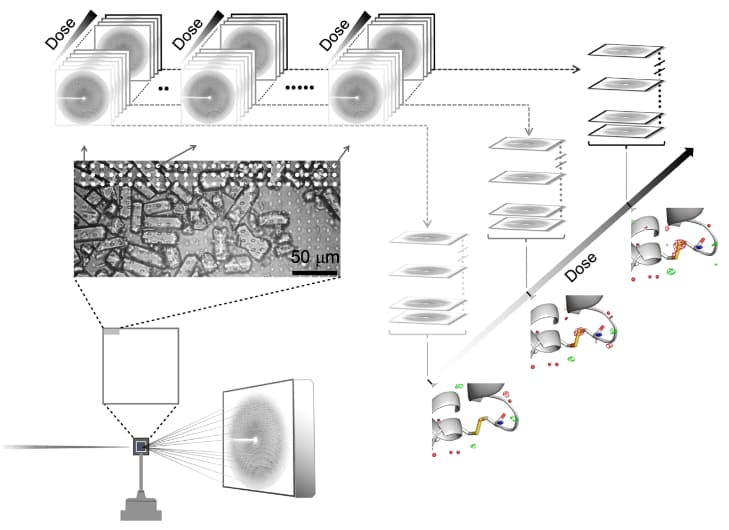
The combination of raster-scanning serial crystallography at ID13 with its highly-brilliant micro-focused X-ray beam and a fast pixel array detector (EIGER X 4M) permitted the splitting of the exposure of a single position on the sample into N individual diffraction patterns, recorded with an exposure time as short as 2 ms per pattern (Figure 1). Such a data-collection series can then be decomposed into N serial crystallography data-sets which can be reduced and refined independently from each other. The resulting N detailed electron density maps can be interpreted as the frames of a 2 ms dose-resolved structural movie revealing specific radiation damage at different sites of the macromolecule (in this case hen egg-white lysozyme).
This study, which has been performed within a collaboration of scientists from the ESRF, the IBS, the ILL, the Universities of Oxford, California San Francisco and Notre Dame, provides insight into the temporal evolution of X-ray radiation damage at room- and cryo-temperatures and at two different dose rates. Modelling the dose-dependent decay of specific and global radiation damage indicates a dose limit of 0.38 MGy should not be exceeded at room temperature. Although this guideline cannot necessarily be generalised to other crystalline proteins, it serves as a yardstick for future room-temperature serial crystallography experiments and helps to define experimental conditions for more detailed radiation damage studies on crystalline proteins. The opportunities which will be offered at fourth-generation synchrotron radiation sources just coming online such as ESRF-EBS should expand the parameter range for room temperature radiation damage studies by at least two orders of magnitude regarding time scale and dose rates, bringing fine-grained time-resolved radiation damage experiments and new biological insights within reach.
Principal publication and authors
E. de la Mora (a), N. Coquelle (b), C.S. Bury (c), M. Rosenthal (d), J.M. Holton (e,f,g), I. Carmichael (h), E.F. Garman (c), M. Burghammer (d), J.-P. Colletier (a), and M. Weik (a), Radiation damage and dose limits in serial synchrotron crystallography at cryo- and room temperatures, Proceedings of the National Academy of Sciences 117, 4142-4151 (2020); doi: 10.1073/pnas.1821522117.
(a) Université Grenoble Alpes, CEA, CNRS, IBS, Grenoble (France)
(b) ILL
(c) Department of Biochemistry, University of Oxford (UK)
(d) ESRF
(e) Department of Biochemistry and Biophysics, University of California, San Francisco (USA)
(f) Division of Molecular Biophysics and Bioengineering, Lawrence Berkeley National Laboratory, Berkeley (USA)
(g) Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory, Menlo Park (USA)
(h) Notre Dame Radiation Laboratory, University of Notre Dame, Notre Dame (USA)