- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Electronic structure, magnetism and dynamics
- Iridium fluoride complexes as a proxy for iridates
Iridium fluoride complexes as a proxy for iridates
Molecular fluoride complexes based on iridium ions are isolated single-metal entities that imitate the iridium sites found in inorganic iridium oxides. XMCD experiments provide evidence of the virtually identical electronic structure of these two iridium systems. These results open a novel bottom-up synthetic approach towards new materials based on heavier transition elements with exotic physical properties.
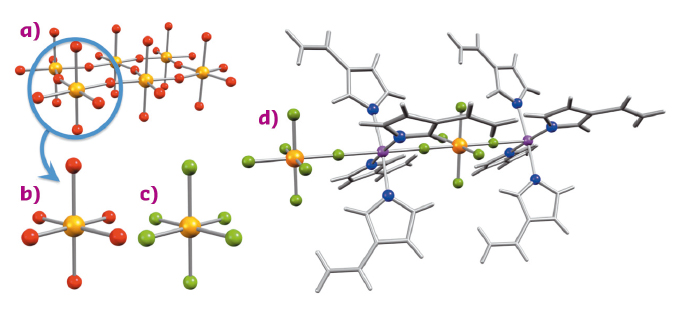
The chemistry and physics of the heavier transition element compounds have been largely overlooked when compared to the lighter first-row transition metals. Several recent key discoveries in the physics of metal oxides based on fourth (4d) and fifth (5d) row transition metals have sparked interest and opened new perspectives for the chemistry and physics of these often overlooked elements [1,2,3]. From recent reports, iridium in particular stands out from the rest. Oxide materials based on Ir4+ ions, commonly referred to as 'iridates', have shown great promise for diverse materials such as high-temperature superconductors, topological insulators and quantum spin-liquids. Their outstanding properties originate from the combined effects of the crystal field, magnetic interactions and strong spin-orbit coupling, characteristic of 5d metal ions. From a fundamental point of view and to understand in detail the origin of these unique properties, the elucidation of the intrinsic electronic structure of the constituent Ir4+ centres is essential. However, this task is far from trivial as the strong magnetic interactions between the iridium ions in iridates eclipse the information of the local electronic structure. To overcome this limitation, the iridate structures (Figure 14a) should be fragmented into its most basic structural unit. Discouragingly, the fundamental structural unit of all iridates, {IrO6}8– (Figure 14b), carries a substantial charge that prevents its spatial isolation in a material with only remotely placed charge-compensating ions. Instead, our attention was recently turned towards a related molecular species, the [IrF6]2– complex (Figure 14c). From a chemical point of view, fluoride and oxide share many characteristics, and, importantly, the reduced charge of the fluoride ion (F–) over oxide (O2–) renders the isolation of a single 'iridate centre' chemically possible. The structure of the [IrF6]2– complex is virtually identical to that of the {IrO6}8– moiety in iridates and there are no indications of disturbing magnetic interactions between adjacent [IrF6]2– complexes, when isolated in the solid state with organic counter-ions. These [IrF6]2–-based materials therefore constitute the most realistic molecular model systems for oxide-based iridates.
 |
|
Fig. 14: a) The IrO4-layer in the well-known Sr2IrO4 structure together with b) the basic structural motif {IrO6}8– and c) its molecular fluoride-based analogue [IrF6]2–. d) View of the linear chain structure of Zn(viz)4[IrF6] (viz = 1-vinylimidazole) incorporating [IrF6]2– as a building block. Colour code: Ir, orange; F, pale green; O, red; N, blue; Zn, purple; C and H, grey sticks. |
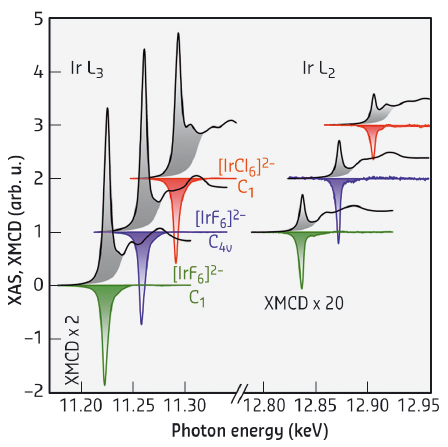
The evidence that the [IrF6]2– complex possesses an electronic structure virtually identical to the {IrO6}8– moiety, came from X-ray magnetic circular dichroism (XMCD) experiments conducted at beamline ID12. Representative spectra collected for different molecular iridate-analogues are shown in Figure 15. Although XMCD experiments are becoming increasingly popular amongst chemists, this technique has not, previously, been applied to molecular 5d metal ion complexes. Remarkably, the modelling of the XMCD data illuminated a large orbital magnetic moment that exceeds the magnetic moment of the electronic spin. The comparison of these results with previous experimental and theoretical studies on iridates demonstrates an almost identical electronic structure of the metal centres in these different types of materials. The [IrF6]2– complex is thus an ideal model system to test hypotheses developed on the single-ion properties of the {IrO6}8– unit in iridates. Moreover, the incorporation of this new [IrF6]2– complex into various chemical environments using the tools of coordination chemistry would allow the influence of different structural modifications on its electronic structure to be probed. In this context, the robust nature of the [IrF6]2– complex is an asset as it facilitates the bottom-up construction of molecule-based materials. This approach is well exemplified by the one-dimensional coordination polymer, Zn(viz)4[IrF6] (Figure 14d), demonstrating the feasibility of designing materials using [IrF6]2– as an 'iridate'-building block. These soft-chemistry methods contrast with the traditional inorganic solid-state chemistry used to synthesise iridates and could be used to obtain new materials with designed topologies and exotic physical properties constructed from [IrF6]2– modules.
 |
|
Fig. 15: Isotropic X-ray absorption (XAS) and XMCD spectra collected at 2 K for different molecular iridate analogues in a large magnetic field of 17 T. |
Principal publication and authors
Iridates from the molecular side, K.S. Pedersen (a,b,c,d), J. Bendix (e), A. Tressaud (c,d), E. Durand (c,d), H. Weihe (e), Z. Salman (f), T.J. Morsing (e), D.N. Woodruff (g), Y. Lan (h), W. Wernsdorfer (h), C. Mathonière (c,d), S. Piligkos (e), S.I. Klokishner (i), S. Ostrovsky (i), K. Ollefs (j), F. Wilhelm (j), A. Rogalev (j) and R. Clérac (a,d), Nat. Commun. 7, 12195 (2016); doi: 10.1038/ncomms12195.
(a) CNRS, CRPP, UPR 8641, Pessac (France)
(b) Univ. Bordeaux, CRPP, UPR 8641, Pessac (France)
(c) CNRS, ICMCB, UPR 9048, Pessac (France)
(d) Univ. Bordeaux, ICMCB, UPR 9048, Pessac (France)
(e) Department of Chemistry, University of Copenhagen (Denmark)
(f) Laboratory for Muon Spin Spectroscopy, Paul Scherrer Institut, Villigen PSI (Switzerland)
(g) Department of Chemistry, University of Oxford (UK)
(h) CNRS, Inst NEEL, Grenoble (France)
(i) Institute of Applied Physics, Academy of Sciences of Moldova, Kishinev (Moldova)
(j) ESRF
References
[1] B.J. Kim et al., Science 323, 1329–1332 (2009).
[2] Y.K. Kim et al., Science 345, 187–190 (2014).
[3] S. Nishimoto et al., Nat. Commun. 7, 10273 (2016).



