- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- X-ray Absorption and Magnetic Scattering
- Liquid Gallium in Confined Droplets Under High Temperature and High Pressure Conditions
Liquid Gallium in Confined Droplets Under High Temperature and High Pressure Conditions
The existence of liquid polymorphism is one of the most interesting and challenging problems in the domain of liquid science to emerge in the last few years. The existence of two distinct liquids, with identical chemical composition but different physical properties, is consistent with experimental data and theoretical studies for a variety of systems. Liquid-liquid phase transitions were related to the soft-core interaction potential widely used to describe liquid metals characterised by the coexistence of metallic and covalent character in the bonding (as liquid Sn, Bi, Ga or Ge). Gallium is a good candidate for showing liquid polymorphism. This is because of its ice-type phase diagram, with a negative temperature dependence of the melting point at low pressure, and because it displays an extended polymorphism in the solid phase. Furthermore, this metal is known to show deep undercooling in the liquid phase at ambient pressure [1].
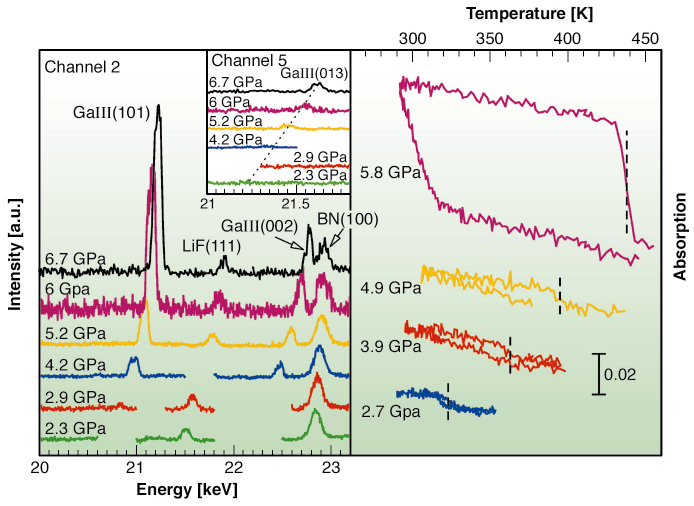
Samples consisting of a dispersion of micrometric gallium droplets (~ 4 % wt) in an inert matrix of mixed epoxy and LiF have been studied by combining Ga K-edge X-ray absorption spectroscopy (XAS), single-energy X-ray absorption detection (SEXAD) and energy-scanning X-ray diffraction (ESXD). The distribution of droplets was fully characterised by scanning electron microscopy (SEM). Measurements were performed at BM29 using a Paris-Edinburgh type large volume press, in a range of pressure and temperature of 0-6.7 GPa and 298-440 K respectively. From ESXD patterns (Figure 109, left panel) the appearance of Ga(III) crystalline phase Bragg peaks is clearly observed above 4.2 GPa (channel 2), whereas no Ga(III) (013) peak is observed at 2.3 GPa (channel 5). The intensity increase of Ga Bragg peaks with increasing pressures indicates that the fraction of crystallised droplets increases with pressure. The appearance of Ga(III) in a region of the pressure-temperature phase diagram where Ga(II) is known to be the stable crystalline phase was previously observed by other works by mean of X-ray diffraction [2] and absorption [3]. In the same figure, right panel, temperature scans (SEXAD) recorded at different pressures present discontinuities associated with the solid-liquid phase transition. The increasing discontinuities observed as a function of pressure, starting from the scan at 2.7 GPa, confirms the occurrence of partial crystallisation of the ensemble of droplets.
 |
|
Fig. 109: Left panel: X-ray diffraction patterns of Ga emulsion collected at RT on channel 2 of the multichannel collimator of the ESXD setup. The trend of the Ga(III) (013) peak as detected on channel 5 for increasing pressures is shown in the inset. The peak is clearly absent below 2.9 GPa. Right panel: temperature scans of the absorption coefficient at constant energy (SEXAD) at various pressures. The melting temperatures are indicated as vertical dashed lines for each loop. |
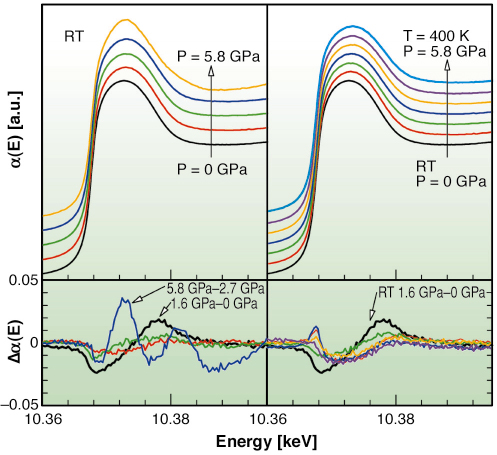
In Figure 110 we report the XAS measurements in an energy interval close to the Ga K-edge for two different thermal histories in the P-T phase diagram. By looking at these spectra and at the corresponding difference signals ![]()
![]() (E), the clear change in the absorption in the liquid phase obtained between 1.6 GPa and 0 GPa may indicate that transformations involving microscopic structure and/or electronic states occur. Also, the shape of the difference signal (black curve) is remarkably different from that obtained for the spectra between 5.8 GPa and 2.7 GPa (relative to the solid and the partially crystallized samples, respectively). It is also different from a typical
(E), the clear change in the absorption in the liquid phase obtained between 1.6 GPa and 0 GPa may indicate that transformations involving microscopic structure and/or electronic states occur. Also, the shape of the difference signal (black curve) is remarkably different from that obtained for the spectra between 5.8 GPa and 2.7 GPa (relative to the solid and the partially crystallized samples, respectively). It is also different from a typical ![]()
![]() (E) curve obtained as a difference between two equilibrium liquid Ga XAS spectra (right panel). This result may infer the existence of a different liquid phase above 1.6 GPa.
(E) curve obtained as a difference between two equilibrium liquid Ga XAS spectra (right panel). This result may infer the existence of a different liquid phase above 1.6 GPa.
 |
|
Fig 110: Upper panels: XAS Ga K-edge spectra as a function of pressure at room temperature (at ambient pressure, at P = 1.6, 1.9, 2.7 and 5.8 GPa), on the left, as a function of pressure and temperature, above the melting curve, on the right. Lower panels: difference signal |
In conclusion, considering both ESXD patterns and SEXAD scans we observe that the quantity of crystallised gallium droplets increases as a function of pressure while no sign of crystallisation is observed up to 2.7 GPa, well beyond the melting point line at room temperature. The exciting hypothesis of liquid-liquid phase transitions occurring between 0 and 1.6 GPa and compatible with the result of this study stimulates further detailed investigations on this system.
References
[1] Di Cicco, Phys. Rev. Lett. 81, 2942 (1998).
[2] L. Bosio, J. Chem. Phys. 68, 1222 (1978).
[3] L. Comez et al., Phys. Rev. B 65, 014114 (2002).
Authors
R. Poloni (a), S. De Panfilis (a), A. Di Cicco (b), G. Pratesi (b), E. Principi (b), A. Trapananti (a) and A. Filipponi (c), Phys. Rev. B 71, 184111 (2005).
(a) ESRF
(b) Università di Camerino (Italy)
(c) Università dell’Aquila (Italy)



