- Home
- News
- Spotlight on Science
- Silk fibre formation:...
Silk fibre formation: using a microfluidic chip to mimic biospinning
26-01-2009
Silk is a biopolymer produced by silkworms and spiders. Its biosynthesis is interesting because of silks remarkable mechanical properties and potential medical applications. In this study, a microfluidic chip was built to mimic the silkworm’s biospinning process. Silk protein aggregation was studied by small-angle X-ray scattering (SAXS) and maturation of the fibre by wide-angle X-ray scattering and Raman spectroscopy.
The conversion of random protein molecules into a fibre by spiders and silk worms is a complex process involving changes in pH, cation concentration and shearing forces. The spinning apparatus consists of a gland connected to a spinneret by a long and tapered duct. At the end of the spinneret, a high-performance semicrystalline fibre exits the animal’s body. There is a great interest in understanding all steps of the silk production line process in order to mimic biospinning. It is, however, practically impossible to study microstructural processes inside the spinning apparatus in situ without harming the animal. Therefore, our work aims at mimicking this process under in vitro conditions, which allow X-ray scattering studies [1].
We have focussed our attention on biospinning by the silkworm Bombyx mori as cocoon silk is a readily available source for fibroin protein. For this purpose, we have built a microfluidic chip with a concentric tube-in-square pipe geometry. The central tube contains the fibroin solution while the surrounding square pipe contains a pH 2 phosphate buffer. Under flow conditions, the diffusional mixing of both solutions allows the pH decrease in the spinning apparatus to be mimicked (Figure 1).
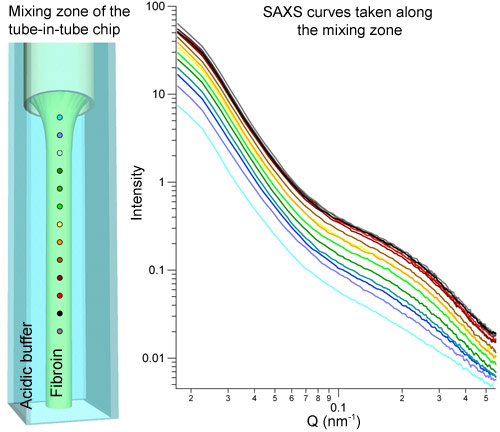 |
|
Figure 1. Mixing zone of the microfluidic chip built to mimic the silk biospinning conditions and SAXS curves obtained during the fibroin aggregation process within this chip. |
Structural changes were probed at selected positions after the onset of mixing by small-angle X-ray scattering at beamline ID02. Modelling of the solution scattering data suggests that the fibroin molecules assemble first into large hollow aggregates (Figure 2). Within these aggregates, the protein molecules adopt a compacted conformation. At the exit of the chip, a silk fibre can be collected in a water bath. After rapid drying, the fibre was probed by wide angle X-ray scattering and Raman spectroscopy at beamline ID13. Interestingly, we produced a fibre rich in random coil and α-helix structures (so-called silk I) (Figure 2), rather than a fibre rich in β-sheet conformation (so-called silk II), which is the end product of the biospinning process. The silk I/silk II conversion could, however, be achieved by increasing the immersion time of the fibre in the water bath [2]; the resulting fibre is shown in Figure 3. These results reinforce a two-step model of fibre formation (Figure 2): an aggregation process followed by conformational and structural maturation. This model has also been proposed for other peptides that form amyloidal conglomerates.
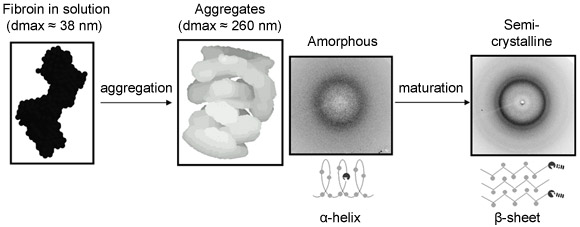 |
|
Figure 2. Aggregation-maturation 2-step model of silk fibre formation. |
 |
|
Figure 3. Microscope image of an artificial silk fibre. |
The present results are an encouraging step towards the complete mimicry and understanding of biospinning. We have, however, not yet produced a fibre showing the high molecular orientation of natural fibres. This will require further development of the microfluidic cell, in particular to mimic shearing forces.
The microfluidic device described here is a general tool to study aggregation processes upon change of chemical environment. The concentric flow condition is an advantage when compared to other microfluidic cells where aggregation at the walls may pose a problem.
References
[1] A. Martel, M. Burghammer, R. Davies, E. DiCola, P. Panine, J.-B. Salmon, and C. Riekel. A microfluidic cell for studying the formation of regenerated silk by synchrotron radiation small- and wide-angle X-ray scattering, Biomicrofluidics 2, 024104 (2008).
[2] A. Martel, Silk protein aggregation in a microfluidic environment, PhD Thesis (2008), http://tel.archives-ouvertes.fr/tel-00322259/fr/
Principal publication and authors
A. Martel, M. Burghammer, R. Davies, E. Di Cola, C. Vendrely, C. Riekel, Silk fiber assembly studied by synchrotron radiation SAXS/WAXS and Raman spectroscopy, J. Am. Chem. Soc. 130, 17070 (2008).
ESRF



