- Home
- News
- Spotlight on Science
- Hydrogen-induced...
Hydrogen-induced Ostwald ripening
11-09-2008
The cluster size of an ensemble of palladium nanoclusters was found to increase on hydrogenation. This phenomenon is attributed to hydrogen-induced Ostwald ripening and originates from the higher mobility of palladium atoms resulting from the low sublimation energy of the palladium hydride when compared to the palladium metal. Future applications of nano-structured hydrogen storage materials will have to take this phenomenon into consideration.
The pursuit of high capacity hydrogen storage materials started long ago and has gained new momentum due to the escalating costs of fossil fuels and our increased awareness of global warming. Hydrogen in combination with a suitable storage medium could be an alternative fuel [1]. The main aim for hydrogen storage materials is to obtain high hydrogen concentrations in a material that possesses suitable transport properties under ambient conditions. A promising way to favourably alter a material's properties is by reducing the size of its grains to the extent that surface and quantum effects begin to play a major role. The investigation of nanocluster metal hydrides may therefore reveal novel properties. Palladium is one of the most widely studied metals with respect to hydrogen absorption. The structural and electronic changes of bulk palladium upon hydrogenation are well documented [2]. In palladium nanoclusters, the hydrogen miscibility gap is reduced considerably as a result of the nanoscale dimensions [3,4]. Moreover, at elevated temperatures palladium nanoclusters change size due to Ostwald ripening [5]: the larger clusters capture mobile atoms at the expense of smaller clusters [6]. As hydrogen in a metal can decrease the strength of the host metal bonding, this raises the question of how an ensemble of palladium nanoclusters will interact during and after hydrogenation. A morphological or structural change of the nanocluster ensemble may affect the hydrogenation properties as compared to a single cluster.
We investigated the effect of hydrogen exposure on a palladium cluster assembled film produced with a dual-target dual-laser vaporisation source [7] with three different techniques: X-ray diffraction (XRD), Extended X-ray absorption fine structure (EXAFS) and scanning tunnelling microscopy (STM). These three complementary methods were used to determine the size changes of the palladium nanoclusters upon exposure to hydrogen and oxygen. The average grain size was derived both from the width of the XRD Bragg peaks and the coordination number around Pd atoms obtained from EXAFS, whereas the (lateral) diameter of the nanoclusters at the sample surface was measured directly by STM. X-ray absorption data collected on the DUBBLE beamline (BM26A) at the ESRF yielded additional parameters such as inter-atomic distances, degree of disorder and electronic information through the shift of the absorption edge; these support the observation of size changes.
The as-deposited clusters have a round, nearly spherical shape with an average diameter of 4.0 nm, which is in good agreement with that measured with the mass spectrometer, indicating that this size is particularly stable. Upon hydrogenation the increase of the nanocluster size measured with XRD, EXAFS, and STM is 22%, 38%, and 37%, respectively. This increase is much larger than the increase of about 8.1% of the Pd phase unit cell volume corresponding to the hydride formation. The cluster growth is due to an atomic re-organisation by three-dimensional Ostwald ripening, whereby the larger clusters take up mobile atoms at the expense of smaller clusters [6]. For most materials, spontaneous Ostwald ripening is an extremely slow process at room temperature. However, the presence of a hydrogen atom in the metal lattice reduces the binding energy, thus increasing the probability of detachment of palladium atoms. Since the clusters are in close contact, the detached atom does not have to travel far to stick to another cluster. This process can be qualitatively understood in terms of the difference between the sublimation energy of palladium metal and palladium hydride. The sublimation energy of palladium decreases with increasing hydrogen concentration and is about 50% lower at a H/Pd ratio of 0.3 [8]. Since our experiment was performed at room temperature, a doubled temperature corresponds to approximately 600 K (327°C). At room temperature Ostwald ripening of palladium clusters is unlikely, while at 300°C larger clusters are formed [9]. Therefore the size increase is attributed to Ostwald ripening induced by exposure to hydrogen: the absorbed hydrogen decreases the sublimation energy of palladium, which stimulates the detachment of atoms from the clusters at room temperature. Hence, an individual nanocluster may have favourable hydrogen storage and transport properties but these may be lost as a result of morphological changes caused by hydrogenation cycles. Since this is very likely a general phenomenon, the exposure of nano-structured materials to hydrogen may need to be viewed from this new perspective.
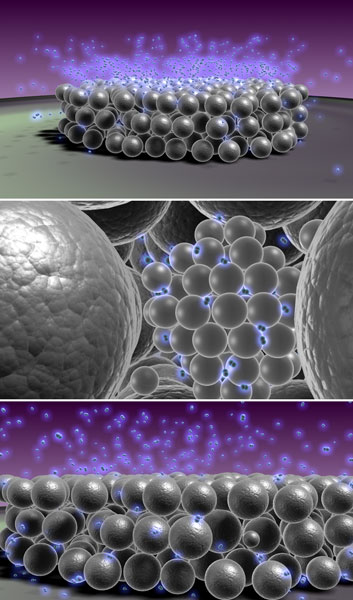 |
|
Figure 1. Schematic impression of hydrogen-induced Ostwald ripening. From top to bottom: Hydrogen atoms approaching the palladium nanocluster assembled film; The palladium atoms within the palladium cluster building blocks become free to move due to the interstitial hydrogen; Some of the clusters become larger at the expense of the smallest. This process is shown in greater detail in the movie: Hydrogen-induced Ostwald ripening at room temperature in a Pd nanocluster film (MPEG, 10 mb). |
References
[1] L. Schlapbach and A. Züttel, Nature 414, 353 (2001).
[2] J. Völkl and G. Alefeld, Hydrogen in Metals I, edited by G. Alefeld and J. Völkl, Topics in Applied Physics, Vol. 28, Springer, Berlin (1978).
[3] J.A. Eastman, L.J. Thompson, and B.J. Kestel, Phys. Rev. B, 48, 84 (1993).
[4] A. Pundt et al., J. Alloys Comp., 293-295, 480 (1999).
[5] A. Howard, C.E.J. Mittchell, and R.G Egdell, Surf. Sci. 515, L504 (2002).
[6] W. Ostwald. Z. Phys. Chem. (Leipzig), 34, 495 (1900).
[7] W. Bouwen et al., Rev. Sci. Instrum., 71, 54 (2000).
[8] N.V. Piskunov et al. , J. Eng. Phys. Thermophys. 74, 1217 (2001).
[9] C.M. Whelan, C.J. Barnes, Appl. Surf. Sci., 119, 288 (1997).
Principal publication and authors
M. Di Vece, D. Grandjean, M.J. Van Bael, C.P. Romero, X. Wang, S. Decoster, A. Vantomme and P. Lievens, Hydrogen-induced Ostwald ripening at room temperature in a Pd nanocluster film, Phys. Rev. Lett. 100, 236105 (2008).
Laboratorium voor Vaste-Stoffysica en Magnetisme, Instituut voor Kern- en Stralingsfysica & INPAC - Institute for Nanoscale Physics and Chemistry, K.U.Leuven (Belgium)



