- Home
- News
- Spotlight on Science
- Locating atoms in...
Locating atoms in a quasicrystal
22-03-2007
Where are the atoms? This question has been puzzling scientists since the discovery of quasicrystals 25 years ago by Shechtman and co-workers [1]. The structure of one of these fascinating materials has just been solved by a team of researchers from Japan and France using the ESRF's X-ray beams.
Quasicrystals are extremely well-ordered structures whose atomic arrangement is nonperiodic. This shows up in their diffraction pattern which displays sharp Bragg reflections (a signature of long range order) but with a 5-fold rotational symmetry, incompatible with lattice translation. Therefore, the powerful tools of standard crystallography cannot be used to determine the structure. A high dimensional crystallographic methodology has been developed, similar to that used for incommensurately-modulated structures. In this approach, periodicity is recovered in a 6-dimensional space, which permits modelling and comparison with experimental data.
The discovery of the icosahedral phase of Cd5.7Yb by Tsai and co-workers [2] was a breakthrough. Indeed this is the first binary quasicrystal, which makes structural analysis much simpler. There is also good contrast for X-ray scattering between the Cd and Yb atoms. Finally, there are two so-called periodic approximants, which can be synthesised with chemical compositions and atomic structure very close to that of the principal quasicrystal. The structure of the approximant crystals is described by a periodic packing of a large structural unit with icosahedral symmetry, whose external shell is a triacontahedron (Figure 1). This forms an atomic cluster which is chemically extremely well ordered, with Yb atoms sitting on the vertices of an icosahedron. The clusters are densely packed and connected along the 2-fold and 3-fold axis, where they interpenetrate.
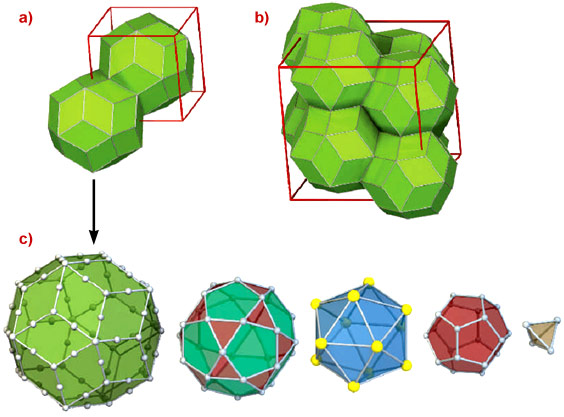 |
|
Fig. 1: Packing of the triacontahedral unit in the 1/1 (a) and 2/1 (b) cubic approximant whose lattice parameter are b=1.57 nm and |
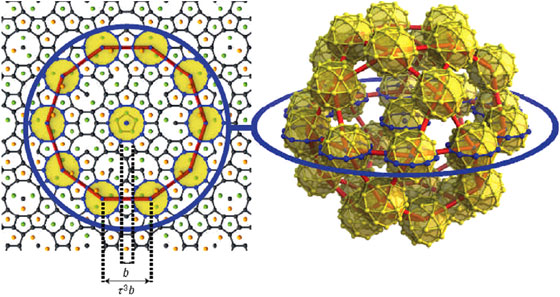
The structure of the icosahedral CdYb quasicrystal has been solved using X-ray diffraction data collected on the D2AM beamline (BM02). Our model was refined against the experimental data by using a 6-dimensional analysis, which included a phase reconstruction procedure, and this in combination with the information obtained from the approximant structure. The large number of Bragg peaks (more than 5000) and their dynamical range (almost eight orders of magnitude) put a severe constraint on the modelling which nonetheless resulted in a very good R factor of 0.096. The CdYb quasicrystalline structure is described as a quasiperiodic packing of the triacontahedral cluster, with connections along 2- and 3- fold axis as observed in the approximant: 94% of the atoms belong to such clusters. Other local environments, unique to the quasicrystal, are also present. The model also leads to a description of the hierarchical packing of the clusters, illustrated in Figure 2: the clusters are packed together to form a 'cluster of clusters' shown as a yellow disk in the left panel. In turn this ‘cluster of clusters’ also forms a larger cluster whose characteristic length is  3 larger, where
3 larger, where  =1.6168 is the golden mean. This inflation property continues at infinity and might be used to explain the quasicrystal’s physical properties.
=1.6168 is the golden mean. This inflation property continues at infinity and might be used to explain the quasicrystal’s physical properties.
 |
|
Fig. 2: Illustration of inflation properties in the quasicrystal. The left panel shows the distribution of cluster centres (black dots) in a 5-fold plane. The yellow disk is a trace of a cluster of clusters shown in the right panel. They form a larger unit as highlighted by the blue circle, shown on both panels. The edge of the left panel is roughly equal to 34 nm. |
Now that this model has been firmly established, it paves the way for further investigation into the stability and physical properties of quasicrystals.
References
[1] D. Shechtman, I. Blech, D. Gratias and J.W. Cahn, Phys. Rev. Lett. 53, 1951 (1984).
[2] A.P. Tsai, J.Q. Guo, E. Abe, H. Takakura and T.J. Sato, Nature 408, 537 (2000).
Principal Publication and Authors
H. Takakura (a), C.P. Gómez (b), A. Yamamoto (c), M. de Boissieu (d), A.P. Tsai (b), Atomic structure of the binary icosahedral Yb-Cd quasicrystal, Nature Materials 6, 58-63 (2007).
(a) Division of Applied Physics, Hokkaido University, Sapporo (Japan)
(b) Institute of Multidisciplinary Research for Advanced Materials, Tohoku University, Sendai (Japan)
(c) Advanced Materials Laboratory, NIMS, Tsukuba (Japan)
(d) Laboratoire de Thermodynamique et de Physico-Chimie Métallurgique, UMR CNRS 5614, ENSEEG-INPG, Saint Martin d’Hères (France)



