- Home
- News
- Spotlight on Science
- Bacterial tubulin:...
Bacterial tubulin: a folding mystery
05-12-2005
Prosthecobacter, a bacterium isolated from raw sewage, may have acquired tubulin during its evolution. Tubulin is essential for higher organisms as it plays a pivotal role in the eukaryotic cytoskeleton and is not normally found in bacteria. The eukaryotic protein polymerises into small fibres called microtubules and forms polar tracks for motor proteins that transport cargo through the cell, such as organelles or chromosomes during cell division.
Using X-rays from ESRF beamlines BM14, ID14-1, ID14-4 and ID29 and crystals of bacterial tubulin we found the similarities between eukaryotic and bacterial tubulin much more pronounced than predicted. While bacterial tubulin still forms fibres and heterodimers, we discovered that the protein can assume proper shape and function in simple buffer solutions from an unfolded state [1]. This seemed impossible because eukaryotic tubulin requires a complicated process to fold, machinery that is not present in bacteria. It remains a mystery why one protein needs many co-factors to assume its shape, while another with a strikingly similar structure can do without.
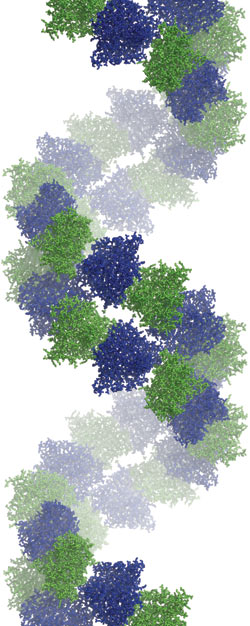
Figure 1 shows the antiparallel double filament with repeating building blocks in the crystal packing, produced by the 6522 space group symmetry. Between the subunits A (blue) and B (green) is a bend of 15° (intra dimer), and between the heterodimeric building blocks is a bend of 45°. In electron micrographs, bacterial tubulin can be seen forming straight or slightly twisted filaments which are similar to protofilaments from eukaryotic tubulin [1].
 |
|
References
[1] D. Schlieper, M.A. Oliva, J.M. Andreu & J. Löwe: Structure of bacterial tubulin BtubA/B: evidence for horizontal gene transfer. Proc. Natl Acad. Sci. USA 102, 9170-9175 (2005).
Authors
Daniel Schlieper and Jan Löwe, MRC Laboratory of Molecular Biology, Cambridge, (UK)



