- Home
- News
- Spotlight on Science
- Untangling the chemical...
Untangling the chemical bonding in high-pressure γ-B28 boron
17-06-2011
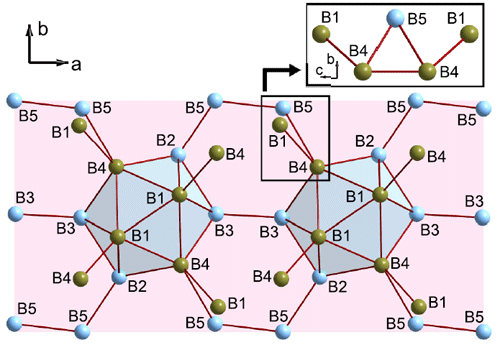
An icosahedral, quasimolecular cluster of 12 boron atoms (B12) is the building block of all allotropes of boron as well as many boron-rich solids. However, the properties of boron polymorphs and boron-rich compounds that make them interesting for materials science and technology (extreme chemical stability associated with high hardness and low compressibility) are highly unusual for molecular solids. An explanation for this phenomenon requires a detailed analysis of the chemical bonding between boron atoms, which scientists have now accomplished for the high-pressure boron polymorph γ-B28.
Modern X-ray diffraction methods permit an insight into the electron density distribution in a material. For this purpose, one would preferably investigate a single crystal of a compound using X-ray synchrotron radiation. Experimental data obtained by X-ray diffraction are analysed with the aim to create electron-density maps which reflect the probability of an electron being present at a specific location. Achieving the required level of accuracy is a demanding task. For the light element boron, this has not been solved so far, mainly because high quality single crystals were unavailable. Recently a reliable method for high-pressure synthesis of boron single crystals was developed at the University of Bayreuth (Bayerisches Geoinstitut), where single crystals of the high-pressure γ-B28 phase were obtained [1, 2].
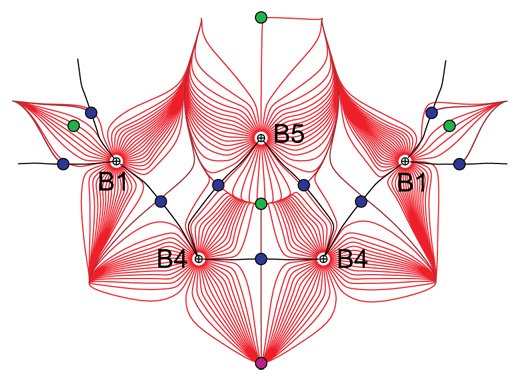
High-resolution, single-crystal X-ray diffraction data of γ-B28 have been collected at a temperature of 85 K at the Swiss-Norwegian beamline, BM01A. Electron-density studies by means of high-quality, low temperature X-ray diffraction data provide important experimental information about chemical bonding in crystalline solids, including the identification of bonding interactions, ionicity vs covalency of bonds, and an estimate of the strength of interactions. A topological analysis of the static electron density has been performed at the Laboratory of Crystallography of the University of Bayreuth according to Baders’ quantum theory of atoms in molecules [3]. The quantum theory of atoms in molecules defines a bond critical point (BCP) as the point of minimum density along the bond path between two atoms (saddle point of the electron density). Similarly, ring critical points (RCPs) characterise closed paths of bonded atoms. Bonding interactions require the presence of a BCP and/or RCP, and the values of the electron density and its Laplacian at BCPs and RCPs correlate with the properties of the chemical bonds. The present experimental static electron density reveals BCPs for all bonds as well as RCPs for three-centre (3c) and polycentre bonds (Figures 1, 2). The arrangement of BCPs and RCPs (Figures 1, 2) is in agreement with the following interpretation of bonding in γ-B28.
Covalent polycentre bonds exist on the B12 clusters. Neighbouring clusters are bonded to each other by two-electron two-centre (2e2c) and electron-deficient, one electron two centre (1e2c) bonds. Strong 2e2c bonds exist within the B2 dumbbells and between the dumbbell and icosahedral groups. Finally, a unique polar-covalent two-electron three-centre (2e3c) bond has been identified between a pair of atoms of one B12 group and one atom of the dumbbell. It is proposed that the charge transfer originates from this peculiar 2e3c bond, to which the three boron atoms contribute unequal amounts of electrons.
The present results explain why boron and boron-rich compounds containing quasimolecular icosahedral B12 clusters acquire physical properties unusual for molecular solids. While these clusters geometrically mimic molecules, intericosahedral chemical bonds are of equal or even higher strength than intraicosahedral bonds, and the structures of boron and boron-rich compounds are controlled by individual two- and three-centre bonds between boron atoms.
Principal publication and authors
Electron deficient and polycenter bonds in the high-pressure γ-B28 phase of boron, S. Mondal (a), S. van Smaalen (a), A. Schönleber (a), Y. Filinchuk (b), D. Chernyshov (b), S.I. Simak (c), A.S. Mikhaylushkin (c), I.A. Abrikosov (c), E.Yu. Zarechnaya (d), L. Dubrovinsky (d), N. Dubrovinskaia (a), Phys. Rev. Lett. 106, 215502 (2011).
(a) Laboratory of Crystallography, University of Bayreuth (Germany)
(b) Swiss-Norwegian Beam Lines at ESRF
(c) Theory and Modeling Division, IFM, Linköping University (Sweden)
(d) Bayerisches Geoinstitut, University of Bayreuth (Germany)
References
[1] E.Yu. Zarechnaya, L. Dubrovinsky, N. Dubrovinskaia, Y. Filinchuk, D. Chernyshov, V. Dmitriev, N. Miyajima, A. El Goresy, H.F. Braun, S. Van Smaalen, I. Kantor, A. Kantor, V. Prakapenka, M. Hanfland, A.S. Mikhaylushkin, I.A. Abrikosov, S.I. Simak. Phys. Rev. Lett. 102, 185501 (2009).
[2] E.Yu. Zarechnaya, N. Dubrovinskaia, L. Dubrovinsky, Y. Filinchuk, D. Chernyshov, V. Dmitriev. J. Cryst. Growth 312, 3388–3394 (2010).
[3] R.F.W. Bader, Atoms in Molecules—A Quantum Theory (Oxford University, New York, 1990).
Top image: A topological analysis of the electron density distribution in γ-B28 boron helps us to understand the unusual physical properties of this quasimolecular solid.