- Home
- News
- Spotlight on Science
- Origin of the bright...
Origin of the bright photoluminescence of silver clusters confined in LTA zeolites
03-09-2018
XEOL-EXAFS was combined with (TD)DFT-based computational modelling and time-resolved optical spectroscopy to unravel the structural and electronic properties of photoluminescent silver clusters in zeolite.
Clusters of a few metal atoms have physical properties distinct from single atoms and larger nanoparticles. Like molecules, they have discrete energy levels allowing electronic transitions conferring remarkable photoluminescent properties [1].
To protect them against aggregation, metal clusters can be stabilised in water-dispersible organic scaffolds such as peptides, proteins, DNA, and polymers or in solid-state matrices such as glasses and zeolites. Zeolites are aluminosilicates found naturally or produced synthetically on an industrial scale and they are used for a wide range of domestic and industrial applications. They consist of a rigid and well-defined crystalline framework made of molecular-scale channels and cavities that are perfectly suited to host small clusters.
Silver clusters that self-assemble in the cavities of faujasite (FAU) and Linde Type A (LTA) zeolites upon heat treatment or X-ray irradiation have homogeneous, photostable and efficient green-yellow emissions reaching unity [2, 3]. This makes them outstanding candidates for light-based applications such as wavelength converters for light-emitting diodes (LEDs) and luminescent tags. Despite their attractive photoluminescence properties, little is known about their structural and electronic properties.
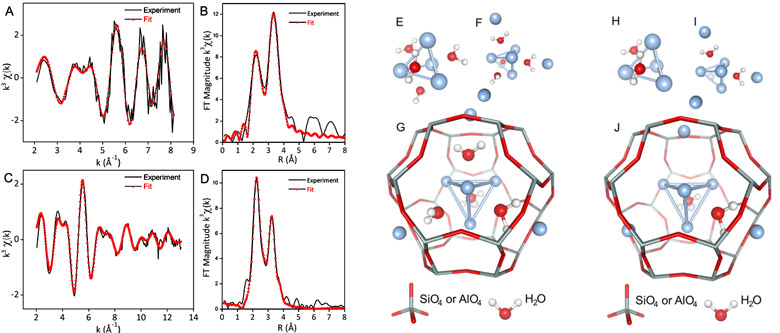
In a new approach, X-ray excited optical luminescence (XEOL)-detected extended X-ray absorption fine structure spectroscopy (EXAFS) was used at beamline BM08 (LISA CRG). XEOL at the Ag K-edge exclusively detects the XAFS signal (Figures 1A&B) from the atoms constituting the emissive species. This innovative technique allowed a selective determination of the structure of the emitting Ag species in LTA zeolite in the presence of charge-balancing K+ ions (Ag3K9-LTA), providing new insight compared to earlier work [3].
The three-dimensional structures of the silver clusters were then determined by combining the XEOL fitting results with those obtained from conventional transmission-detected EXAFS (Figures 1C&D) collected at beamlines BM08 (LISA CRG) and BM26A (DUBBLE CRG). With this technique, the luminescence of silver LTA zeolites was unambiguously assigned to a mixture of two doubly-charged four-atom tetrahedral hydrated silver clusters [Ag4(H2O)x2+ (x = 2 and 4)] positioned at the centre of sodalite cages of the LTA zeolite (Figures 1E to J).
The structures obtained experimentally were confirmed computationally with geometry optimisations by using density functional theory (DFT) methods, while time-dependent DFT (TD-DFT) was applied to calculate the optical transitions. This theoretical modelling indicates that the remarkable optical properties resulting from this structure originate from a confined super-atom quantum two-electron system in which hybridised silver and water oxygen orbitals are highly delocalised over the cluster.
Upon excitation, one electron of the s-type highest occupied molecular orbital (HOMO) 1S0 is promoted to the p-type lowest unoccupied molecular orbitals (LUMO) 1P, perturbed by the water ligand field interaction, and relaxes through enhanced system intercrossing into green luminescent long-lived microsecond triplet states 3P as presented in Figure 2. These calculated transitions were confirmed by time-resolved optical spectroscopy.
This study may be extended to other materials containing few-atom clusters made of silver and, perhaps, other metals. This new-found understanding may yield an entire class of materials with tailored properties, based on luminescent silver clusters, that should find technological applications in the near future.
A summary of this research is presented in the animation: Why silver clusters captured in zeolites emit light: https://youtu.be/SAVkW9Lm80M
Principal publication and authors
Origin of the bright photoluminescence of few-atom silver clusters confined in LTA zeolites, D. Grandjean (a), E. Coutiño-Gonzalez (b), N.T. Cuong (c,d), E. Fron (b), W. Baekelant (b), S. Aghakhani (a), P. Schlexer (e), F. D’Acapito (f), D. Banerjee (g), M.B.J. Roeffaers (h), M.T. Nguyen (d), J. Hofkens (b), P. Lievens (a), Science 361, 686–690 (2018); doi: 10.1126/science.aaq1308.
(a) Laboratory of Solid State Physics and Magnetism, KU Leuven (Belgium)
(b) Molecular Visualization and Photonics, KU Leuven (Belgium)
(c) Faculty of Chemistry, Hanoi National University of Education (Vietnam)
(d) Department of Chemistry, KU Leuven (Belgium)
(e) Dipartimento di Scienza dei Materiali, Università di Milano-Bicocca (Italy)
(f) BM08-LISA beamline, ESRF (France)
(g) BM26A-DUBBLE beamline, ESRF (France)
(h) Centre for Surface Chemistry and Catalysis, KU Leuven (Belgium)
References
[1] H. Hakkinen, Adv Phys-X 1, 467-491 (2016).
[2] G. De Cremer et al., J Am Chem Soc 131, 3049-3056 (2009).
[3] O. Fenwick et al., Nat Mater 15, 1017-1022 (2016).
![Frontier orbitals of [Ag4(H2O)4(Si24H24O36)]2+ and energy level diagram of Ag4(H2O)22+ and Ag4(H2O)42+ clusters in Ag3K9-LTA.](/files/live/sites/www/files/news/spotlight/2018/spotlight323/spotlight323-Fig2sm.jpg)



