- Home
- News
- Spotlight on Science
- Revealing Old Masters...
Revealing Old Masters pigment grades by synchrotron X-ray diffraction
05-04-2018
Two complementary X-ray diffraction techniques were used to analyse historical paint micro-samples from paintings by Leonardo da Vinci and Jean de Beaumetz. This work provided insight into the various grades of lead white pigments used by the Old Masters, revealing valuable information for curators, conservators and art historians.
Lead white was one of the first pigments synthesised by man. In ancient times, it was obtained through a complex manufacturing process in which metallic lead sheets were exposed to an atmosphere containing acetic acid vapour, CO2, O2 and H2O. Pigment manufacturers were able to induce lead corrosion, thus obtaining the white pigment containing a mixture of crystalline lead carbonates: cerussite PbCO3 and hydrocerussite 2PbCO3.Pb(OH)2 [1]. This synthesis of lead white was used until the 19th century, when a alternative routes were adopted for pigment production. From this unique synthesis process, one might have expected the continuous production of an identical material over time. However, historical sources reveal that different qualities of lead white were sold to artists through pigment markets at very different prices and painters were able to select different grades of pigment for use in their artworks [2]. Numerous post-synthesis processes could be used by the manufacturers, such as an acid treatment (ground in vinegar) or heating in water. The best products were praised for their exceptional optical and handling properties. While all the recipes were carefully recorded in ancient painting treatises, the Old Masters never revealed what type of lead white they used in their artworks.
By using a combination of two synchrotron X-ray diffraction (XRD) configurations at different beamlines, this research has revealed the grades of lead white within micro-samples (< 100 µm) collected from masterpieces of French collections, notably those of the Louvre and Orsay Museums.
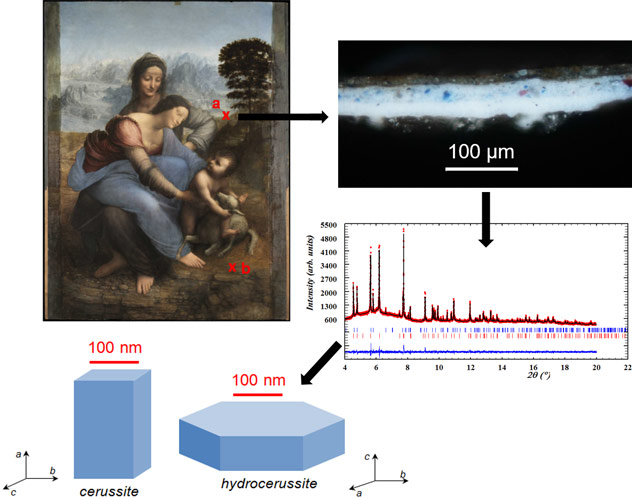
Laboratory reconstructions of ancient synthesis processes and of post-synthesis treatments permitted an investigation of their effects on the composition and microstructure of the pigment. For example, heating the pigment in water induced the transformation of cerussite into hydrocerussite, with the newly formed hydrocerussite crystallites of large sizes (dimensions up to 5 µm instead of < 1 µm without this process). Conversely, the treatment with acidic led to the formation of tiny (< 100 nm) cerussite crystallites, following a recrystallisation process.
At beamline ID22, micro-samples were sealed into glass capillaries, and rotated in a 35 keV beam, to avoid preferential orientation effects. High-angular resolution XRD (SR-HXRD) was then combined with Rietveld refinement to obtain quantitative phase compositions with high precision (± 2 w%) and crystallite morphologies at the nanometric scale [3]. These results were compared to samples obtained through reconstructions of the ancient treatments, allowing an explanation for the measured hydrocerussite to cerussite ratios and crystallite sizes. For example, the lead white pigment used by Leonardo da Vinci (1452-1519) in his masterpiece La Vierge, l’Enfant Jésus et Sainte Anne (1503-1519) is characterised by a high amount of cerussite, and crystallites of nanometric sizes, that might result from treatment in vinegar (Figure 1).
At beamline ID21, high-spatial resolution XRD (SR-µXRD) permitted crystalline maps to be obtained at the micrometric scale on complex multi-layered samples. Historical micro-samples were scanned using a sub-micrometric 8.5 keV beam (0.7 x 1.0 µm2): at each point of analysis, a diffractogram was collected and fitted. The technique permitted discrimination between the various lead whites used within the same painting, within superimposed layers of micrometric thickness. It was possible to identify a specific type of lead white pigment in a painting by Jean de Beaumetz (c.1335-1396) by comparison of the obtained structural data with that from historical sources. Beaumetz was known to be working for Philip the Bold at the Duchy of Burgundy, and all the materials he used were carefully recorded in accounting books that have luckily been conserved until today. Those accounts reported the purchase of two white pigment types, at different prices: blanc de plomb (lead white) and blanc de puille (puille white). The precise nature of blanc de puille was unknown to art historians: the only certitude is that it cost half the price of blanc de plomb. The study of crystalline phase maps collected at ID21 revealed the existence of a pigment composed of 100% hydrocerussite, with crystallites of thickness ~400 µm, used in a very thin layer (Figure 2). It is now suspected that this very specific composition corresponds to blanc de puille, and could result from a treatment in hot water, that might have been used to clean a low-grade pigment, in order to get rid of tarnishing impurities.
Principal publication and authors
Synchrotron-based high angle resolution and high lateral resolution X-ray diffraction: Revealing lead white pigment qualities in Old Masters paintings, V. Gonzalez (a,b,c), G. Wallez (b,c,a), T. Calligaro (a,b), M. Cotte (d,e), W. De Nolf (d), M. Eveno (a), E. Ravaud (a), and M. Menu (a,b), Analytical Chemistry 89, 13203-13211 (2017); doi: 10.1021/acs.analchem.7b02949.
(a) Centre de Recherche et de Restauration des Musées de France, C2RMF, Palais du Louvre, Paris (France)
(b) PSL Research University, Chimie ParisTech - CNRS, Institut de Recherche de Chimie Paris, UMR8247, Paris (France)
(c) Sorbonne University, UPMC University, Paris (France)
(d) ESRF
(e) Laboratoire d’Archéologie Moléculaire et Structurale (LAMS), Sorbonne University, UPMC University, Paris (France)
References
[1] E. Welcomme et al., App. Phys. A 89, 825 (2007).
[2] M. Stols-Witlox et al., in The Artist’s Process, Archetype Publications (2012): 112.
[3] V. Gonzalez et al., Microchem. Journal 125, 43 (2015).