- Home
- News
- Spotlight on Science
- Observing new ways...
Observing new ways to stack colloidal cubes
10-02-2017
The self-organisation of colloidal cubes leads to the creation of novel ordered structures with fascinating optical properties. Optical microscopy combined with ultra-small-angle X-ray scattering data collected at beamline BM26 (DUBBLE) has uncovered new ways to stack colloidal cubes and their rich phase behaviour.
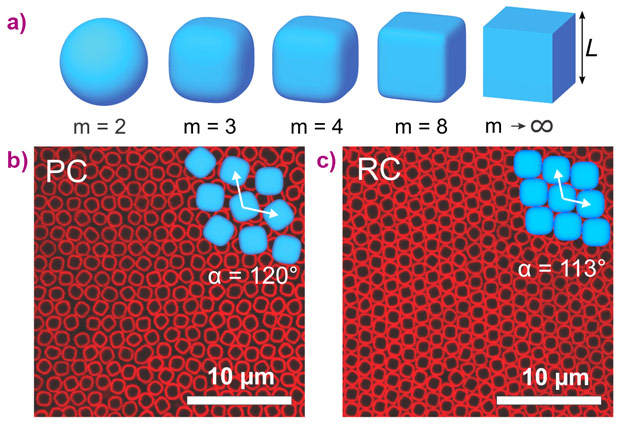
Colloids are able to spontaneously self-organise into ordered colloidal crystals with periodicity comparable to the wavelength of visible light leading to remarkable optical properties such as a photonic bandgap. In the vast majority of studies, spherical colloids have been used, which are known to form close-packed structures such as face-centred cubic or (random-stacking) hexagonal close-packing. However, by varying the particle shape, one can obtain structures with very different symmetries [1]. Subsequent to the recent synthesis of cubic colloids in our laboratory [2], we can study the spontaneous self-assembly of cubic building blocks. Due to slightly rounded corners, these particles can be remarkably well described by the superball formula: |x|m + |y|m + |z|m = (L/2)m, where m is the shape parameter. With m = 2 it describes a sphere with a diameter L, and for m → ∞ the shape approaches a perfect cube (Figure 1a). Surprisingly, simulation studies show that the phase behaviour of these superballs does not result in simple cubic structures but instead show a phase transition from plastic crystals to rhombic crystals [3]. Here, we studied the assembly of hollow silica cubes experimentally and uncovered new ways to stack colloidal cubes and their rich phase behaviour.
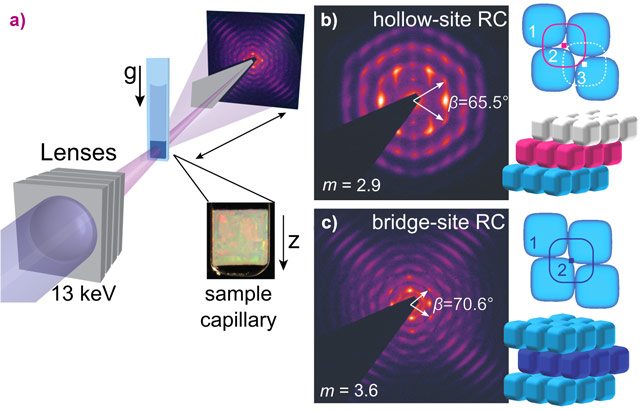
The colloidal suspensions were placed in capillaries that were stored vertically. Gravity induces a monotonic increase of the osmotic pressure towards the bottom of the capillary allowing the observation of various coexisting phases within one sample. By using a combination of confocal laser scanning microscopy (CLSM) and small-angle X-ray scattering with microradian resolution (µrad-SAXS), insights into the ordering process of superball colloids were obtained. CLSM experiments showed the formation and transition from a plastic crystal phase, with positional order and orientational disorder (Figure 1b), into a fully-ordered rhombic crystal with orientational order (Figure 1c). The high angular resolution of the microradian setup [1] at beamline BM26 allowed the investigation of the 3D structures of crystals of these micron-sized colloids (Figure 2a). The 2D µrad-SAXS patterns showed the presence of large monocrystalline regions with rhombic symmetry, where the stacking sequence of the planes was found to be dependent on the exact shape of the superball particles. For m < 3.2, a hollow-site stacking sequences was found (Figure 2b), while for m > 3.4, a unique bridge-site stacking sequence was observed (Figure 2c).
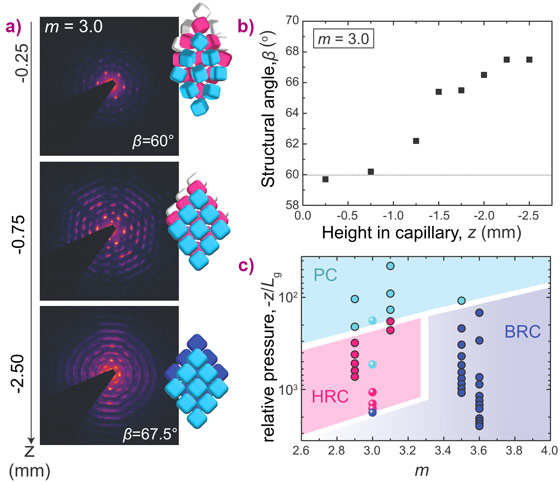
In addition, the µrad-SAXS revealed the effects of the osmotic pressure on the crystal structures. For superballs with slightly ‘softer’ interactions, the transition from a plastic crystal to a rhombic crystal with hollow-site stacking was surprisingly followed by a second transition into bridge-site stacking at the highest osmotic pressure (Figure 3a). The first transition is characterised by an increase of the structural angle β from hexagonal (β = 60°) to rhombic (β > 60°) (Figure 3b). This observation of solid-solid transitions involving at least three distinct polymorphic structures is unique for colloids. Based on all investigated superball shapes and pressures, an experimental phase diagram was constructed (Figure 3c).
Thus, colloidal cubes were observed to crystallise into plastic crystals followed by distinctly different rhombic crystal structures, where the stacking variant, hollow-site or bridge-site, was determined by the exact superball shape and osmotic pressure. Our findings partly confirm the predicted phase behaviour but find a richer phase behaviour with the observation of the hollow-site stacking. These findings bring us one step closer to ultimately controlling the experimental self-assembly of superballs into functional materials, such as photonic crystals.
Principal publication and authors
Observation of solid-solid transitions in 3D crystals of colloidal superballs, J.-M. Meijer (a), A. Pal (a) S. Ouhajji (a), H.N.W. Lekkerkerker (a), A.P. Philipse (a) and A.V. Petukhov (a,b), Nature Communications 8, 14352 (2017); doi: 10.1038/NCOMMS14352.
(a) Van 't Hoff Laboratory for Physical and Colloid Chemistry, Utrecht University (The Netherlands)
(b) Laboratory of Physical Chemistry, Eindhoven University of Technology (The Netherlands)
References
[1] A.V. Petukhov et al., Curr. Opinion Colloid Interf. Sci., 20, 272–281 (2015); doi: 10.1016/j.cocis.2015.09.003.
[2] L. Rossi et al., Soft Matter, 7, 4139-4142 (2011); doi: 10.1039/c0sm01246g.
[3] R. Niet al., Soft Matter, 8, 8826-8834 (2012); doi: 10.1039/c2sm25813g.