- Home
- News
- Spotlight on Science
- Degradation of magnesium...
Degradation of magnesium implants in bone: mechanisms of Mg storage and impact on bone structure
01-03-2016
Advanced X-ray microbeam techniques at the ESRF have enabled a team of Austrian researchers to study the reaction of bone to biodegradable bone implants based on Mg. The study unveiled an impact of the implant degradation speed on the Mg distribution, indicating at least two different pathways of Mg storage in bone. Changes in bone nanostructure and mineralisation were observed in a region of high local Mg content. The results suggest direct incorporation of Mg in bone mineral crystals.
While conventional bone implants were developed to minimise corrosion and interaction with the body, a novel class of biodegradable magnesium implants has been designed to support the bone as long as needed and then to be dissolved and replaced by bone tissue as soon as healing has sufficiently progressed. In this approach, controlling the degradation kinetics is imperative for successful treatment. While the implant corrosion process for Mg and the surgical aspects are fairly well researched, the fate of the degradation products and their mid- to long-term impact on the bone nanostructure and mineral composition was still unclear. Such questions are, however, of high scientific interest and key issues for clinical use.
Bone is known as a hierarchically structured material with a highly optimised architecture from the macroscopic down to the nanometre level, where bone is a nanocomposite of hydroxyapatite (HAP) mineral platelets embedded in a collagen fibre matrix. The nanostructure plays an important role in determining the mechanical properties and reflecting the integrity of healthy bone. Since bone can adapt to changing external influences, the impact of implant placement and degradation on its structure and mineralisation is an important puzzle piece in understanding the healing process. In particular, the expected very high local Mg concentration due to implant degradation has the potential to influence bone structure and mineralisation.
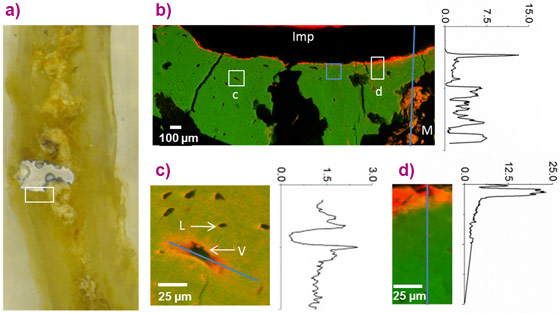
A combination of scanning µSAXS (micro small-angle X-ray scattering) and µXRD (micro X-ray diffraction) with a spatial resolution of 3 µm was carried out at beamline ID13 along with further µXRF (micro X-ray fluorescence) experiments at ID21, which allowed the distribution of Mg in the bone matrix to be examined with a spatial resolution of about 500 nm.
Figure 1a shows a rat femur with the regions of synchrotron investigation highlighted by rectangles. The degradation after 3 months is quite advanced owing to the fast degradation speed of the alloy [1]. A clear tendency to accumulate Mg at the bone-implant interface as well as an accumulation around blood vessels can be seen. The Mg content at the interface and in the blood vessel regions was estimated to be as high as 15% and 2.5% respectively, considerably above the usually reported content of 0.5% for the natural level of Mg in bone.
Interestingly, the accumulation at the interface occurred in the case of fast implant degradation, when the high local availability of Mg apparently exceeded the natural buffer capacity of the blood vessel system. In contrast, in the case of slow degradation, the usual site of Mg accumulation is around the blood vessel. Further experiments showed a normalisation of the Mg accumulation around the blood vessel back to the normal Mg background at approximately 12 months after complete implant degradation [2].
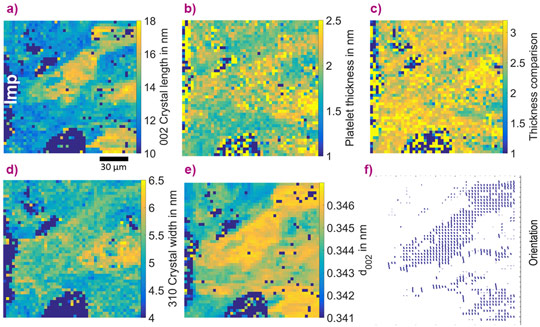
µXRD and µSAXS at beamline ID13 on the same samples unveiled changes in the nanostructure and mineral composition due to the presence of Mg. Figure 2a and d show the lengths and widths of the hydroxyapatite crystals respectively as fitted from the Scherrer broadening of the 002 and 310 peaks. The analysis of the mineral platelet thickness and orientation from the SAXS signal, shown in Figure 2b and f, shows a distinct pattern of orientation, higher mineral platelet thickness and longer and thicker crystals in regions of low Mg content. In regions of high Mg content, the mineral platelets are smaller, shorter and less thick and the c-axis of HAP, as denoted by the 002 d-spacing in Figure 2e, is markedly contracted, indicating lattice distortion. As shown in Figure 2c, the ratio of SAXS/XRD thickness is larger in areas with higher Mg content, possible evidence for an amorphous layer at the outside of the mineral platelets, consisting of HAP with a lattice distorted by Mg inclusion. Further experiments on the laboratory scale showed the onset of realignment of the HAP platelets and a subsequent normalisation over the course of 12 months [3].
In conclusion, this study helped in the understanding of the transport, storage and incorporation mechanisms of unusually high local Mg levels in bone. The findings suggest a preferential accumulation of Mg around blood vessels and in the bone marrow in contrast to the initial hypothesis of Mg accumulation at the bone-implant interface. The interface could be identified as the storage site for Mg at very high degradation and Mg release rates. The long-term data indicate a normalisation of the Mg levels after sufficiently long healing time, suggesting only temporary storage in the bone matrix. The mineral composition and nanostructure appears to be temporarily altered by the presence of Mg as the HAP c-axis is contracted and platelets are formed which are markedly shorter and thinner.
This study highlights the importance of matching the degradation behaviour of Mg implant alloys with the physiological needs of healing bone. We expect that a thorough understanding of the underlying processes in bone will greatly advance the development of improved degradable implant materials for the benefit of patients of all ages, but specifically children that run the risk of bone deformation in the case of permanent implants and currently often have to undergo a second surgical intervention for implant removal.
Principal publication and authors
Magnesium from bioresorbable implants: distribution and impact on the nano- and mineral structure of bone, T.A. Grünewald (a), H. Rennhofer (a), B. Hesse (b), M. Burghammer (b,c), S.E. Stanzl-Tschegg (a), M. Cotte (b), J.F. Löffler, (d), A.M. Weinberg (e), H.C. Lichtenegger (a), Biomaterials 76, 250-260 (2016); doi: 10.1016/j.biomaterials.2015.10.054.
(a) Institute of Physics and Materials Science, University of Natural Resources and Life Science (BOKU), Vienna (Austria)
(b) ESRF
(c) Department of Analytical Chemistry, Ghent University (Belgium)
(d) Laboratory of Metal Physics and Technology, Department of Materials, ETH Zurich (Switzerland)
(e) Department of Orthopaedics, Medical University of Graz (Austria)
References
[1] T. Kraus et al., Acta Biomaterialia 8, 1230-1238 (2012).
[2] T.A.Grünewald et al., Biomaterials 76, 250-260 (2016).
[3] T.A. Grünewald et al. Acta Biomaterialia 31, 448-457 (2016).
Top image: µXRF concentration map of Mg and P in rat bone at the immediate interface to the degrading Mg implant.