- Home
- News
- Spotlight on Science
- Bone tissue mass...
Bone tissue mass density is determined by the canalicular network morphology
07-11-2014
Phase-contrast based X-ray imaging techniques at the nanoscale were used by a team of European researchers to gain new insight into mineral exchange at the interface between the bone-cell network and the surrounding mineralised matrix of human bone tissue.
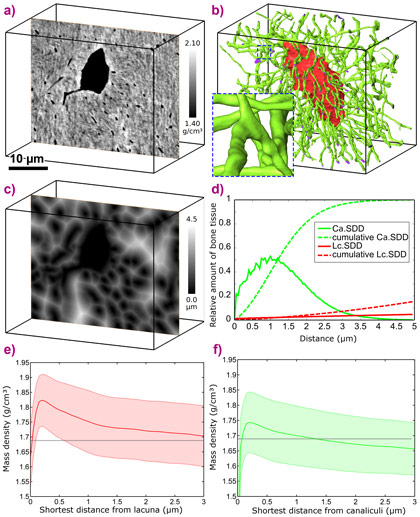
Bone is a biological material structured hierarchically over several length scales, from the molecular level of collagen to the organ level. Bone quality in terms of strength depends on bone properties at all length scales. Details on the morphology of the micro/nano porosity as well as the properties of the surrounding mineralised tissue at this scale are of high interest to understand bone function. In bone remodelling, maturation of the newly formed osteonal bone tissue is associated with a rapid primary increase in mineralisation followed by a slower secondary phase. This increase in mineralisation requires supply and precipitation of minerals into the bone matrix. Mineral delivery can occur only from the extracellular fluid via interfaces such as the Haversian system (housing the blood vessels) and the osteocyte pore network. Osteocytes, of which there are as many as tens of thousands per cubic millimetre of bone, are housed in small cavities called lacunae, and are connected via an extensive network of dendrites that are housed in small canals, called canaliculi, only few hundreds of nm in diameter (Figures 1 and 2). In the present study, we hypothesised that mineral exchange during mineralisation is achieved by the diffusion of minerals from the lacunocanalicular network to the bone matrix, resulting in a gradual change in tissue mineralisation with respect to the distance from the pore-matrix interface. Based on this assumption, alterations in the mass density distribution with tissue age would be expected. In addition, we hypothesised that mineral exchange occurs not only at the lacunar, but also at the canalicular boundaries.
Synchrotron radiation phase-contrast nano computed tomography experiments were carried out at beamline ID22NI (now ID16A). We can now show evidence that the mass density in the direct vicinity of the lacunocanalicular network is indeed different from the mean mass density of the bone tissue, resulting in mass density gradients with respect to both the lacunar and the canalicular boundaries (Figure 2). The results indicate that mass density gradients diminish with increasing tissue age, resulting in a higher and a more homogeneously distributed extra-cellular matrix mass density in old tissue. These findings support our hypothesis that extra-cellular matrix mineralisation is achieved through the diffusion of calcium from the extracellular fluid contained by the lacunocanalicular network to the bone matrix through all interfaces. The smaller mass density values and gradients found in the peri-canalicular tissue compared to the peri-lacunar regions can be explained by the morphology of the lacunocanalicular network, which indicates a higher mineral flux at the lacunar interfaces. We estimate that the amount of calcium stored in the sub-domain of the bone tissue located within 0.5 µm distance from the lacunocanicular network greatly exceeds the amount required for a rapidly exchangeable calcium pool to be used to compensate daily fluctuations in the serum calcium level. However, further studies are needed to confirm a mineral flux from the mineralised matrix into the lacunocanalicular pore network. Supporting the important role of this micro-pore network, we quantified the surface density of the lacunocanalicular network to be about 20 times higher than the surface density of the Haversian system. This relation holds despite the volume ratio of the lacunae being approximately one order of magnitude smaller than that of the Haversian canals.
In line with recent studies, these findings underline the importance of the entire lacunocanalicular network in mineral homeostasis and the influence of its morphology on the spatial distribution of mass density in the mineralised extracellular matrix of bone. The role of the increased mass density adjacent to this pore network should be taken into account in drug deposition studies and mechanical models studying the mechanosensation of osteocyte. Future studies using synchrotron radiation phase-contrast nano computed tomography will aid in the investigation of the diffusion and mineralisation processes and their coordination via osteocytes or other factors.
Principal publication and authors
Canalicular network morphology is the major determinant of the spatial distribution of mass density in human bone tissue - evidence by means of synchrotron radiation phase-contrast nano-CT, B. Hesse (a,b), P. Varga (b), M. Langer (a,c), A. Pacureanu (a,c,d), S. Schrof (b), N. Männicke (b), H. Suhonen (a), P. Maurer (e), P. Cloetens (a), F. Peyrin (a,c), K. Raum (b), Journal of Bone and Mineral Research, in press (2014), DOI: 10.1002/jbmr.2324.
(a) ESRF
(b) Berlin-Brandenburg School for Regenerative Therapies & Julius Wolff Institute, Charité, Universitätsmedizin Berlin (Germany)
(c) Université de Lyon, CREATIS; CNRS UMR5220; Inserm U1044; INSA-Lyon; Université Lyon 1 (France)
(d) Centre for Image Analysis and Science for Life Laboratory, Uppsala University (Sweden)
(e) Klinik für Mund-, Kiefer- und Gesichtschirurgie; Klinikum Bremerhaven-Reinkenheide (Germany)
Top image: Surface representation of the lacunar (red) and canalicular (green) for a region containing one osteocyte lacuna.