- Home
- News
- General News
- How do breast milk...
How do breast milk components interact with human norovirus?
09-05-2016
Norovirus, a highly contagious stomach bug, affects millions of people of all ages around the world. There are very few ways to prevent infections and the disease can be dangerous for babies, since they can quickly become dehydrated. Researchers have shown, thanks to work on several beamlines at the ESRF, that components of breast milk may block norovirus from attaching to cellular ligands that are necessary for an infection. This is the first structural study that shows the binding interactions at the atomic level.
New parents spend the first months of their child’s life stressed about multiple aspects of parenthood, including dealing with illnesses that can affect their babies. Norovirus causes about 1 in 5 cases of gastroenteritis worldwide (according to the Center for Disease Control and Prevention in the US) and it is highly contagious. Noroviruses are transmitted via the fecal-oral route and many infections often result from consumption of contaminated foods such as clams and oysters. There are many genetically distinct types of noroviruses, so people can get infected numerous times throughout their lives.
Human noroviruses bind to the so-called Histo-Blood Group Antigens (HBGAs), which can be found on epithelial cells - a potential target cell for human norovirus. At least nine different HBGA types have been found to interact with human noroviruses. On the other hand, the third most abundant component of human breast milk, Human Milk Oligosaccharides (HMOs), also contains similar building blocks that structurally resemble those of HBGAs. HMOs can resist degradation in the gut and the majority is excreted intact in the feces. This implies that HMOs can travel the same path as norovirus in the host. Previously, a few studies had indicated that human milk’s oligosaccharides could interact with the norovirus capsid, but the binding sites on the particles were not known.
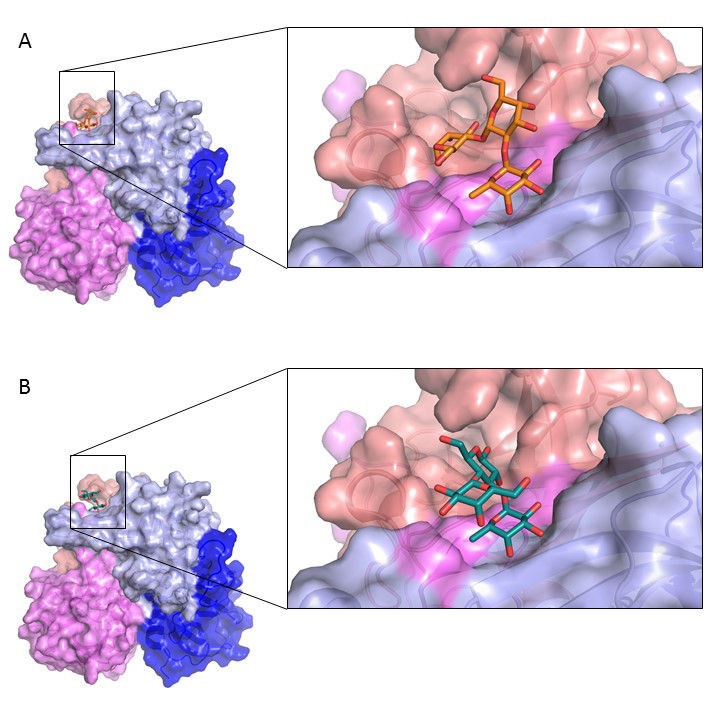 |
|
Surface representation of a norovirus P domain in complex with (A) 2’FL (orange sticks) and (B) 3FL (teal sticks). The norovirus P dimer was coloured according to (chain A and B) and P1 and P2 subdomains, i.e., chain A: P1 (blue), chain A: P2 (light blue), chain B: P1 (violet), chain B: P2 (salmon). Superposition and close-up of the various orientations of the HMOs and citrate bound to the P dimer. Credit: G. Hansman. |
Researchers from Heidelberg University (supported by the Chica and Heinz Schaller Foundation), the German Cancer Research Center, and the company Jenneweln Biotechnologie GmbH have now analyzed the ability of human norovirus to bind to two oligosaccharides from human breast milk: 2’-fucosyllactose (2’FL) and 3-fucosyllactose (3FL). This time, the ESRF played a major role in collecting the X-ray diffraction data of the norovirus spike protein in complex with these two HMOs. The team found that both 2’FL and 3FL were capable of blocking norovirus binding to HBGA samples. The experiments took place on beamlines ID23-1, ID30A-1 (MASSIF-1) and ID30B. It was the first published result of an experiment by users on ID30B. Christoph Mueller-Dieckmann, the beamline responsible, explains the advantages of the newest addition in the beamline portfolio in this research: “A highly intense, stable beam in conjunction with the possibility to screen many samples is crucial for such an experiment”.
Potential applications from this study include the option of using 2’FL and 3FL as norovirus antivirals. Today, 2’FL is already used as a safe food supplement for infant formula, but the potential for norovirus antiviral therapy has not yet been explored. Dr. Grant Hansman, from the German Cancer Research Center/ Heidelberg University is hopeful about the future: “Clinical trials with 2’FL and 3FL are expected in the very near future”.
Reference:
Weichert S., et al, doi:10.1128/JVI.03223-15.
Text by Montserrat Capellas Espuny



