- Home
- News
- General News
- A key protein of...
A key protein of the Sleeping Sickness keeps scientists awake
14-01-2019
Scientists from Germany have discovered how an inhibitor against an essential protein from the parasite causing African sleeping sickness works. Every year up to 11000 people in the sub-Saharan region are infected causing around 3.500 deaths. The new findings could help in the future design of drugs combatting this disease.
“Completely unexpected and extremely interesting!”, this is how Ute Hellmich, Professor at the Johannes Gutenberg-University Mainz (Germany) and the Centre for Biomolecular Magnetic Resonance (BMRZ) of the Goethe-University Frankfurt, describes the moment when she and her team saw that a covalent inhibitor targeting an essential enzyme in African trypanosomes led to the dimerization of the protein – something they did not foresee.
African trypanosomes, the causing agent for African sleeping sickness – a disease classified by the WHO as a neglected tropical disease - are transmitted by the Tsetse fly. Trypanosomes also infect and kill cattle resulting in severe economic repercussions in already impoverished countries. The parasite involved in African sleeping sickness is very similar to those causing Chagas disease in South America and Leishmaniosis worldwide.
While there are some treatment options available against African sleeping sickness, patients have to deal with severe side effects. “Ultimately we’d like to come up with drugs that only affect the parasite, not the humans, so that they can be effective without detriment to the patient’s health”, explains Hellmich.
 |
|
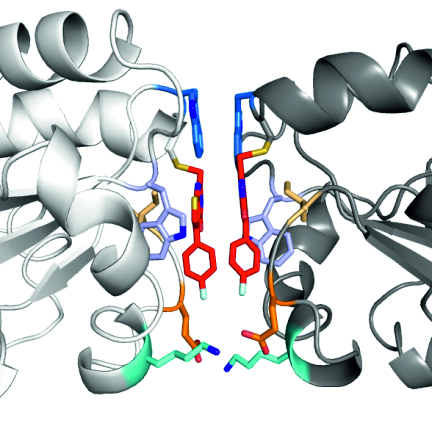
The perfect dimer: Crystallographically observed inhibitor-mediated dimerization interface. Credits: Wagner, A. |
The team of scientists from the University of Mainz, the BMRZ Frankfurt, the University of Würzburg, the University of Heidelberg and the EMBL came to the ESRF to see how a known inhibitor of the tryparedoxin (Tpx) oxidoreductase essential to the life cycle of the parasite binds to its target. The scientists’ original aim was to obtain a high-resolution X-ray crystal structure of the inhibitor-Tpx complex, but they did not count on the inhibitor to induce stable dimerization of the protein. Intriguingly, this requires intricate interactions between different parts of the dimer, both between the inhibitor moieties and the protein monomers.
Perfect dimer
“Our first experiments on ID23 and ID29 led to a high resolution crystal structure with a stable dimer. Wanting to know more and convinced that it was a crystallization artefact, we went to BM29, where we carried out small-angle X-ray scattering (SAXS) experiments to double-check that what we had seen in the crystal also occurs in solution. And there it was, the dimer! We would never have predicted what we saw, we were all very excited”, says Hellmich.
“This is a very nice example showing how the complementary techniques available at the ESRF can be used together”, says Gordon Leonard, head of the ESRF Structural Biology group. “Having the possibility to use SAXS to confirm the inhibitor-induced dimerization of Tpx has helped to push this project more quickly than it might otherwise have been the case”, he adds.
An interesting aspect of this research is the fact that a very small molecule is able to efficiently induce protein dimerization. In most other examples where a molecule glues together two proteins, two connected “arms” of the same molecule (which is often rather large) bring the proteins together. In this research though, the team used a tiny molecule, which binds covalently to a single protein, and only when two proteins, each with a bound inhibitor, meet, do they form a stable dimer.
“Our research describes a new way how an inhibitor interacts with a protein which may be exploited in future drug design efforts. This research is very fundamental and was only possible through a highly interdisciplinary approach. Although I think it will be some time before new drugs without side effects are available, this is a good start”, says Hellmich. “Our next step is to continue our investigations by studying similar parasites causing other tropical illnesses”, she concludes.
Reference:
Text by Montserrat Capellas Espuny
Top image: The tsetse fly, who can carry the parasite that infects humans and animals. Credits: International Atomic Energy Agency.



