- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2017
- Structure of materials
- A step towards organic zeolites
A step towards organic zeolites
Hexacoordinate silicon is often seen in molecular compounds, but very rarely in crystalline silicate materials. Now, reversible Si-O chemistry has been used to assemble octahedral dianionic SiO6 building units and anthracene derivatives into crystalline microporous silicate organic frameworks that share characteristics of both covalent organic frameworks and inorganic zeolites.
Microporous materials are used extensively in adsorption, catalysis, separation and energy storage and production. The field of microporous materials has been dominated for a long time by zeolites. The combination of net charges (and extra-framework cations) and the well-defined crystalline framework structure of these natural and synthetic aluminosilicates are of utmost importance for their successful application as catalyst, adsorbent and ion-exchanger.
Nevertheless, there are still many applications that cannot be addressed by inorganic zeolites. A major synthetic challenge has recently been overcome with the discovery of two other classes of microporous crystalline frameworks, metal-organic frameworks (MOFs) [1] and covalent organic frameworks (COFs) [2]. These materials illustrate that microporous crystalline framework materials incorporating organic building blocks can be achieved. MOFs are hybrid 3D frameworks constructed from metal clusters and multitopic organic linkers assembled via metal-ligand coordination bonds. COFs are purely organic microporous extended crystalline frameworks composed solely of organic linkers linked through strong covalent bonds, thus holding promise for a generation of much more robust materials.
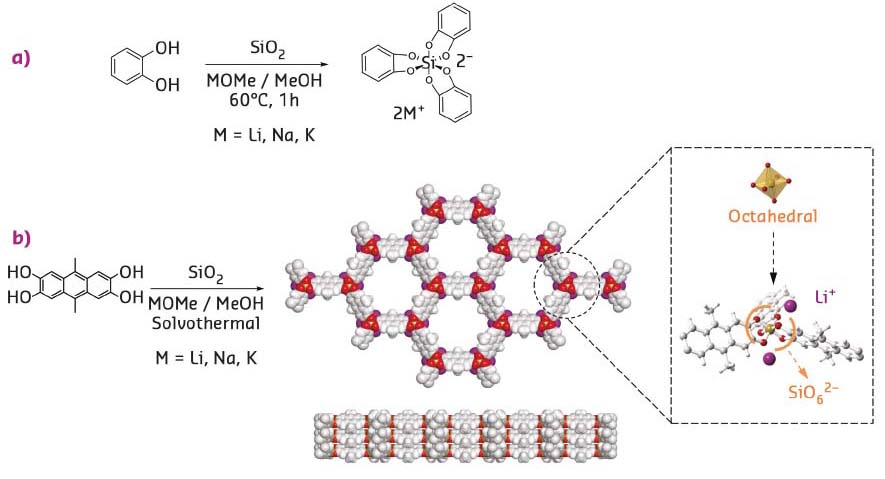
In this study, the synthesis of anionic covalent organic frameworks that bridge the gap between inorganic zeolites and COFs is reported. The frameworks were designed by exploiting the ability of silicon to form hypercoordinate compounds and by implementing reversible Si-O chemistry for the crystallisation of organic building blocks into extended crystalline frameworks. One critical step prior to the formation of a COF based on a new type of linkage consists in identifying a model compound that forms under reversible conditions and that simultaneously fulfils the criteria for the formation of extended crystalline frameworks. Catechol (1,2-dihydroxybenzene) is known to dissolve silica under basic conditions to form tris(catecholato) silicate dianions with octahedral geometry and in quantitative yields (Figure 117a), providing a non-polymeric compound for reference. The formation of silicates is reversible under such conditions and the synthesis of negatively charged silicate covalent organic frameworks (SiCOFs) with alkali-metal counter-ions (Li, Na, K) was successfully carried out in a simple one-pot procedure by condensation of silica and anthracene derivatives under solvothermal alkaline conditions (Figure 117b).
 |
|
Fig. 117: a) Model condensation reaction of catechol and SiO2 used to form tris(catecholato) silicate compounds. b) Anionic silicate covalent organic framework structure (top and side view) and formation by extending the reaction to an anthracene derivative. |
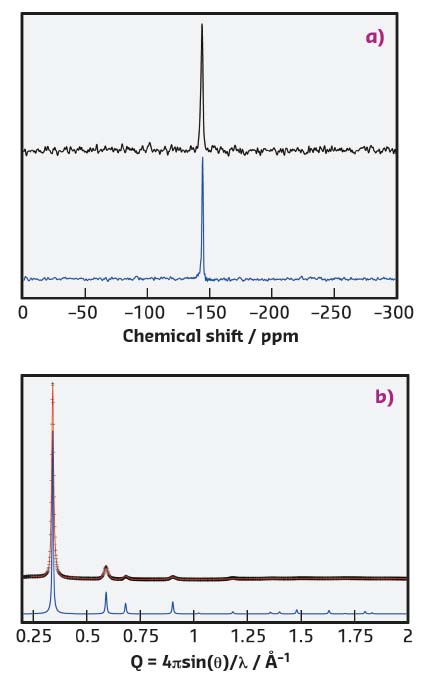
Clear evidence of the hexacoordinate environment around the silicon atoms was given by 29Si nuclear magnetic resonance analysis. A single peak was observed in the 29Si spectrum showing a good match with the one observed for the tris(catecholato) silicate model compound, indicating the presence of solely hexacoordinate dianionic silicon species within the network with no sign of defect (Figure 118a).
 |
|
Fig. 118: a) Comparison of the 29Si CP-MAS solid-state NMR spectra of SiCOF (top) and the tris(catecholato) silicate model compound (bottom) proves that solely hexacoordinate silicon species are present within the framework. b) High-resolution powder X-ray analysis of SiCOF-Li with the observed pattern in black (λ = 0.399977 Å), the Pawley refined profile in red (Rwp = 4.40%) and the theoretical PXRD pattern in blue. |
Such frameworks are synthesised as polycrystalline powders, which makes their structure solution challenging. High-resolution PXRD patterns and high-energy PXRD patterns suitable for analysis by pair-distribution function were collected at beamline ID22 and revealed, in combination with density functional theory calculation, that SiCOFs crystallise as two-dimensional hexagonal layers stabilised in a fully-eclipsed (AA) stacking arrangement with pronounced disorder in the stacking direction (Figures 117b and 118b). Microporosity with high surface area (up to 1300 m2·g–1) was evidenced by gas-sorption measurements and showed that the theoretical maximum surface area was nearly reached experimentally.
This work demonstrates that crystalline silicate material based on hexaoxosilicon units can form under mild reaction conditions by simple reaction of silica as a silicon source. Indeed, in silicate materials, silicon sites adopt almost exclusively a tetrahedral coordination with respect to oxygen and hexaoxosilicon centres within crystalline silicate materials have only been reported for a few high-pressure silicate phases, such as the silica polymorph stishovite.
Principal publication and authors
Anionic silicate organic frameworks constructed from hexacoordinate silicon centres, J. Roeser (a), D. Prill (b), M. J. Bojdys (c), P. Fayon (d), A. Trewin (d), A. N. Fitch (e), M. U. Schmidt (b) and A. Thomas (a), Nature Chemistry 9, 977-982 (2017); doi: 10.1038/nchem.2771.
(a) Department of Chemistry, Technische Universität Berlin (Germany)
(b) Institute of Inorganic and Analytical Chemistry, Goethe University, Frankfurt am Main (Germany)
(c) Department of Organic Chemistry, Charles University in Prague (Czech Republic)
(d) Department of Chemistry, Lancaster University (United Kingdom)
(e) ESRF
References
[1] H. Furukawa et al., Science 341, 1230444 (2013).
[2] C. Diercks et al., Science 355, eaal1585 (2017).



