- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Complex systems and biomedical sciences
- Optimised phase-contrast imaging method slashes radiation dose in mammography
Optimised phase-contrast imaging method slashes radiation dose in mammography
A new approach to X-ray phase contrast imaging developed at the ESRF enables unprecedented low doses in breast imaging and has the potential for being translated into clinical use.
Since the emergence of X-ray phase-contrast imaging (XPCI) more than two decades ago, breast imaging has been considered as one of the applications with the highest potential impact. Conventional attenuation-based X-ray imaging methods are limited in the soft tissue contrast they can achieve, and thus in the discrimination between healthy and cancerous tissues. Additionally, the radiation dose needs to be kept as low as possible due to the high radiosensitivity of the breast. As a consequence, mammography has shortcomings in both sensitivity and specificity, leading to undetected malignancies and unnecessary biopsies, respectively. XPCI provides highly-enhanced soft-tissue contrast. Besides, higher X-ray energies can be used since X-rays do not need to be absorbed in tissue to generate contrast, potentially enabling dose reduction strategies. If implemented in the clinics, dose reduction and image quality improvement would be extremely beneficial, as they would make mammography both a safer and a more accurate exam for breast cancer detection.
Impressive contrast enhancements have been demonstrated in breast imaging through XPCI [1]. However, researchers have struggled to obtain significant dose reductions with respect to conventional mammography, with most studies actually reporting doses exceeding clinical limits [1]. Here, we set out to demonstrate that an optimised approach to XPCI can lower the dose in mammography by at least one order of magnitude compared to clinical practice. To reach this aim, we exploited a combination of cutting-edge developments in novel detector technologies, XPCI and image processing.
 |
|
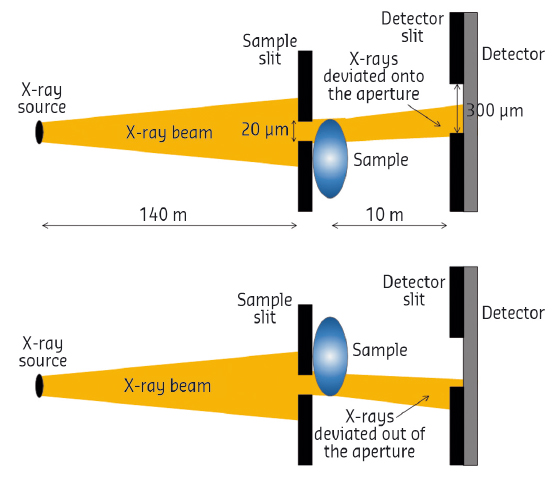
Fig. 116 : Schematic diagram of the EI XPCI setup (not to scale). |
Edge illumination (EI) XPCI was chosen due to its high phase sensitivity [2] and low-dose capabilities [3]. This technique is based on the use of two absorbing slits, placed before the sample and detector respectively, slightly misaligned with respect to each other (Figure 116). In this way, tiny refraction angles are converted into a measurable intensity modulation, as they change the fraction of photons reaching the detector [2]. So far, to retrieve absorption and phase maps of the sample, two EI images needed to be acquired at different slit misalignments and combined [2,3]. In this work, a new retrieval method based on the acquisition of a single image was used [4], which exploits the near homogeneous composition of the breast by assuming the sample absorption and phase to be proportional. Besides simplifying the acquisition procedure, the use of a single-image algorithm enables dose reduction and faster implementations of EI.
A high X-ray energy (60 keV) was chosen to reduce the radiation dose to the sample. At this energy, however, the efficiency of most detectors is limited. Therefore, the MAXIPIX detector, developed at the ESRF [5], was employed. Due to its photon-counting capabilities and its detection efficiency close to 100% at this energy, MAXIPIX is able to minimise image noise for a given X-ray fluence.
 |
|
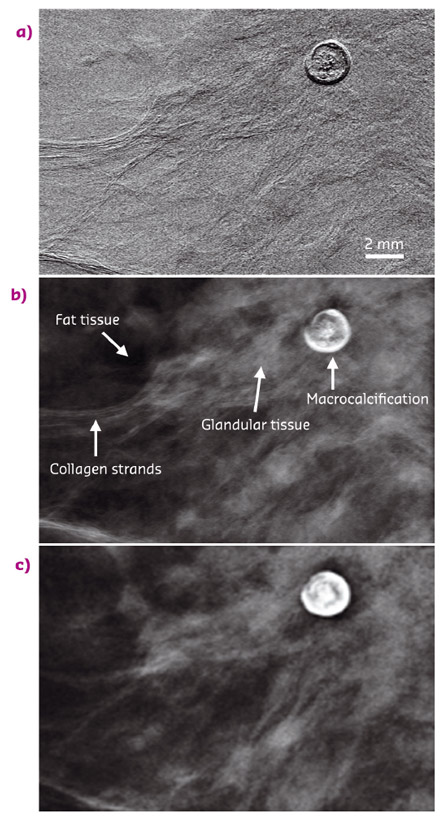
Fig. 117: Images of a 2 cm thick breast sample: a) EI raw image, b) retrieved phase image, c) conventional mammogram. |
Images of excised breast samples of 2-4 cm thickness were acquired at beamline ID17 (The Biomedical Beamline), with a mean glandular dose of only 0.12 mGy. In Figure 117, the raw EI image (Figure 117a) and the retrieved phase image (Figure 117b) are compared to the conventional mammogram acquired at a hospital (Figure 117c) (32 kVp, dose of 4.1 mGy). Despite the high X-ray energy and the extremely low radiation dose, all the breast structures are highlighted in the raw EI image by the refraction-induced signal present at their boundaries. The area contrast typical of conventional X-ray images (more easily interpretable to radiologists), in turn, is restored in the retrieved phase map. This image depicts breast features with at least the same quality as in the conventional mammogram, although at a dose reduced by more than one order of magnitude.
The EI method has also been demonstrated to work efficiently in table-top setups exploiting conventional rotating anode sources [3]. The next step in our research will thus focus on the translation of these considerable dose reductions to a laboratory EI setup, as this would enable the method to be implemented in a clinical setting. Several steps are still needed, including the identification of suitable X-ray sources, beam filtration and optimum system geometry, and the design of larger masks able to cover the entire size of the breast.
Principal publication and authors
A method for high-energy, low-dose mammography using edge illumination X-ray phase-contrast imaging, P.C. Diemoz (a), A. Bravin (b), A. Sztrókay-Gaul (c), M. Ruat (b), S. Grandl (c), D. Mayr (d), S. Auweter (c), A. Mittone (e), E. Brun (b,e), C. Ponchut (b), M.F. Reiser (c), P. Coan (c,e) and A. Olivo (a), Phys. Med. Biol. 61, 8750-8761 (2016); doi: 10.1088/1361-6560/61/24/8750.
(a) Department of Medical Physics and Biomedical Engineering, University College London (UK)
(b) ESRF
(c) Institute for Clinical Radiology, Ludwig-Maximilians University, Munich (Germany)
(d) Institute for Pathology, Ludwig-Maximilians University, Munich (Germany)
(e) Department of Physics, Ludwig-Maximilians University, Garching (Germany)
References
[1] P. Coan et al., J. Phys. D: Appl. Phys. 46, 494007 (2013).
[2] P.C. Diemoz, et al., Phys. Rev. Lett. 110, 138105 (2013).
[3] C.K. Hagen et al., Med. Phys. 41, 070701 (2014).
[4] P.C. Diemoz et al., J. Synchrotron Radiat. 22, 1072-7 (2015).
[5] C. Ponchut et al., J. Instrum. 6, C01069 (2011).



