- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- X-ray nanoprobe
- Speciation of phosphorus bearing inorganic phases in surface sludge deposit of a vertical flow constructed wetland
Speciation of phosphorus bearing inorganic phases in surface sludge deposit of a vertical flow constructed wetland
In order to better describe P species within a sludge deposit from a constructed wetland, four complementary methods were performed: sequential P extractions, 31P solution RMN, X-ray fluorescence (XRF) mapping and X-ray absorption near edge structure (XANES) spectroscopy at P and Fe K-edge. XRF mapping and XANES spectroscopy allowed the different P-bearing Fe- and Ca-species to be distinguished, contributing to our understanding and crucial for the prediction of the potential reversibility of phosphorus capture within the complex organic environmental samples under study.
Phosphorus (P) may be considered both as a beneficial fertilising element and a pollutant since its release into surface aquatic ecosystems may induce eutrophication. P treatment from domestic wastewaters has therefore received increasing attention in recent years. A particular combined wetland treatment process has been developed [1] for the treatment of domestic wastewaters. The process includes a biological trickling filter followed by dephosphatation with ferric chloride dosing. The coagulated suspended solids are then separated from effluent and progressively accumulate at the constructed wetland surface to form a P-rich sludge deposit (above 2% w/w of P in dry matter [2]).
Several complementary analytical techniques were used for qualitative and quantitative descriptions of P species within sludge samples taken from a real municipal wastewater treatment plant. Sequential extractions provided quantitative information on the distribution of P in organic and inorganic fractions. Solution 31P NMR spectroscopy showed the presence of orthophosphates monoesters, diesters and phosphonates. Inorganic P species were identified using a combination of X-ray fluorescence (XRF) mapping and X-ray absorption near edge structure (XANES) spectroscopy at P and Fe K-edge at beamline ID21 to accurately locate the different types of P-bearing phases and determine their status.
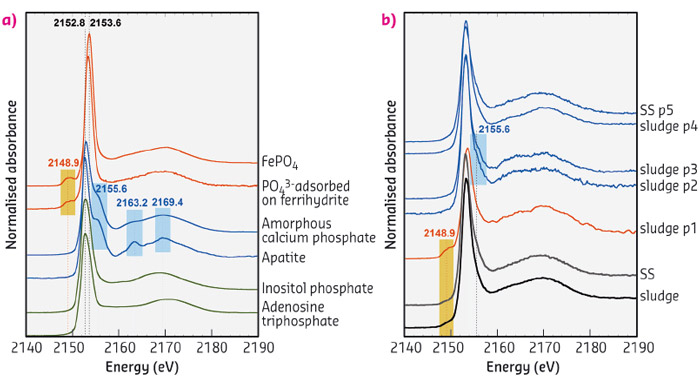
The spectra collected on suspended solids and sludge pellets were found to be very similar to each other (Figure 22b). The pre-edge around 2148.9 eV attested to the presence of phosphates-FeIII bonds (see FePO4 and PO43– sorbed onto ferrihydrite, Figure 22a). Moreover, the broad enlargement of the white line around 2155.6 eV attested also for the presence of phosphates-Ca bonds (see the post-edge shoulder and oscillations at 2163.2 and 2169.4 eV for calcium phosphate in Figure 22a). Spectra collected with the µ-beam on P-rich points of Interest (POIs; p1 to p5) confirmed the presence of FeIII-species (p1 spectrum). Comparison with collected and literature reference spectra [3,4] showed that p2 to p5 were close to amorphous calcium phosphate, phosphate sorbed onto calcite or dicalcium phosphate. The oscillation around 2163.2 eV remained however systematically less pronounced in the spectra collected on suspended solids and sludge samples than in reference spectra.
 |
|
Fig. 22: P K-edge XANES spectra of (a) a selection of reference P minerals (blue and red lines) and organic P species (green lines) and (b) the sludge and suspended solids (SS) samples, collected using an unfocused beam (bottom) and on selected points of interest (p1 to p5) using a µ-beam (top). |
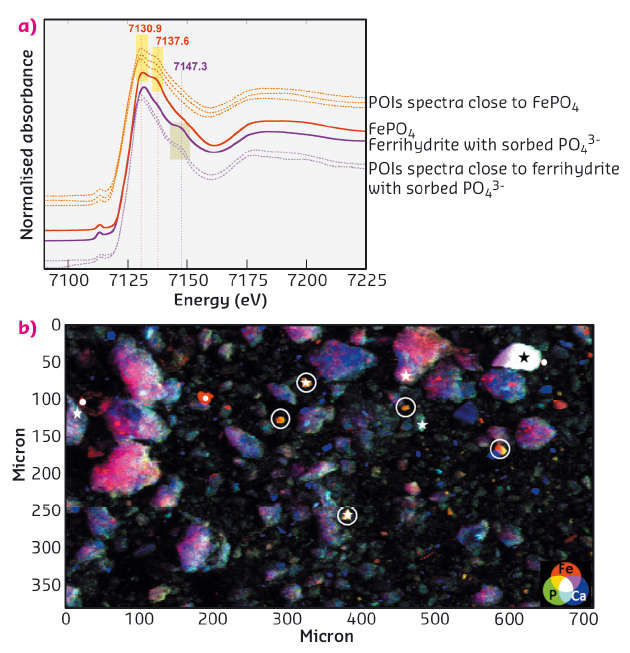
The discrimination between FePO4 and phosphates sorbed onto ferrihydrite remained difficult from spectra collected at P K-edge. However, their corresponding XANES spectra at Fe K-edge evidenced distinct features with an additional peak at 7137.6 eV for FePO4 in comparison to phosphates adsorbed onto ferrihydrite, and an additional peak at 7147.3 eV for ferrihydrite in comparison with FePO4 (Figure 23a). The shape of XANES spectra and elemental compositions of POIs obtained from Fe K-edge µ-XRF maps (Figure 23b) evidenced that P was predominantly bound to iron in forms of ferric phosphate, rather than sorbed onto ferric oxyhydroxide. In addition, the Fe K-edge bulk spectrum collected from sludge sample was well-fitted by a linear combination of FePO4, ferrihydrite and Fe3O4 spectra (with respective contribution of 52, 30 and 19 %), confirming the predominance of FePO4 among Fe-bearing species.
 |
|
Fig. 23: (a) Fe K-edge XANES spectra of FePO4 and phosphates sorbed onto ferrihydrite (references, bold lines) compared with μ-XANES spectra on POIs of sludge sample (dotted lines). (b) µ-XRF map collected above Fe K-edge with points of interest (POIs) having corresponding spectra close to FePO4 (stars) and ferrihydrite with sorbed phosphates (filled circles). |
By combining sequential P extractions, 31P solution RMN and XANES spectroscopy, the present study provided good descriptions of P species within organic environmental complex samples. XRF mapping and XANES spectroscopy allowed us to distinguish the different P-bearing Fe- and Ca-species, contributing to a better understanding of the P retention mechanisms. The generated knowledge was thus crucial to predict the potential reversibility of P capture within the surface sludge deposit.
Principal publication and authors
Effect of aging on phosphorus speciation in surface deposit of a vertical flow constructed wetland, B. Kim (a,b), M. Gautier (a), C. Rivard (c), C. Sanglar (d), P. Michel (b) and R. Gourdon (a), Environmental science and technology. 49, 4903-4910 (2015); doi: 10.1021/es506164v.
(a) INSA Lyon, University of Lyon, LGCIE-DEEP, Villeurbanne (France)
(b) SCIRPE, Sainte-Foy-Lès-Lyon (France)
(c) ESRF
(d) Université de Lyon, CNRS, Institut des Sciences Analytiques, Villeurbanne (France)
References
[1] B. Kim et al., Ecological Engineering 71, 41-47 (2014).
[2] B. Kim et al., Water Science and Technology 68, 2257-2263 (2013).
[3] C. Giguet-Covex et al., Geochimica et Cosmochimica Acta. 118, 129-147 (2013).
[4] J. Rajendran et al., Materials science and engineering C, 33, 3968−3979 (2013).



