- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structure of materials
- Formation of quasi-solid structure by adsorbed noble gas atoms in pores of metal-organic framework
Formation of quasi-solid structure by adsorbed noble gas atoms in pores of metal-organic framework
In situ high-resolution synchrotron powder diffraction measurements of noble gas adsorption in metal-organic framework CuI-MFU-4l revealed a stepwise filling of adsorption sites: 10 crystallographically different positions for Xe and 8 for Kr. Formation of a quasi-solid structure with very dense packing of adsorbed noble gas atoms was observed above the boiling temperature of the corresponding noble gases.
The development of porous materials for selective adsorption of the heavier noble gases has attracted much attention over last few years, being driven by the technological requests for noble gas-storage, separation, and sensing [1]. Xenon, in particular, is an expensive gas that has a lot of potential applications such as in lighting, thermal insulation, excimer lasers, ionisation chambers, general anaesthesia, and NMR probes. The very low concentration of Xe in the atmosphere requires energy-intensive cryogenic distillation of large volumes of air, and gives a mixture of Xe and Kr. The cryogenic separation of Xe/Kr mixtures in rare gas production remains a critical step that could be improved by sorption-based separation technologies. Other applications include Xe recovery from spent nuclear fuel and storage of radioactive Kr.
We have studied the unique noble gas adsorption and storage properties in the exceptional thermally and chemically stable metal-organic framework CuI/-MFU-4l, which contains alternating large and small voids [2]. Measurements at various pressures and temperatures revealed very high volumetric uptake of Xe and Kr as well as a unique quasi-solid noble gas structure in the pores of CuI-MFU-4l, which was formed above the boiling temperature of the corresponding gases.
The high-resolution powder diffraction data collected at beamline ID22 allowed precise localisation of crystallographically different adsorption positions, 10 for Xe atoms and 8 for Kr atoms. Surprisingly, the main adsorption site for both Xe and Kr is not located near an open metal site in the small void, rather it is located in the corner of the large void and interacts with the largest part of the framework. For both Xe and Kr atoms, the coordinatively unsaturated CuI sites are not the first occupied adsorption sites, and their filling starts only after the major adsorption site near the framework in the large void has been filled.
The formation of the first adsorption layer by noble gas atoms in CuI-MFU-4l is complete after filling 4 different adsorption sites in the large pores and one adsorption site in the small pores. Further increase of pressure (or decrease of temperature) results in the formation of a second adsorption layer in the large pores, which consists of 3 positions. The formation of the second adsorption layer in the small pores (2 positions) was observed only for Xe atoms, showing that small and large pores are not equivalent for noble gas adsorption.
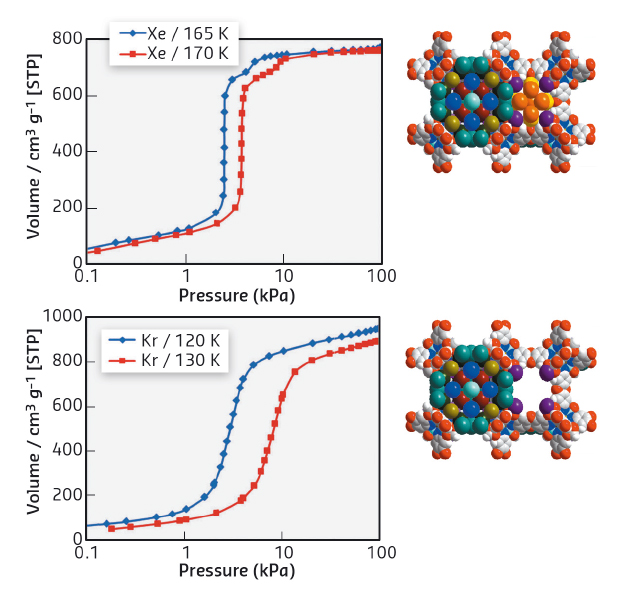
The filling of accessible voids in the small pores is responsible for the inflection in the Xe adsorption isotherms (Figure 41, top). It can be seen that the main adsorption step for Xe adsorption is unusually steep and reflects highly-selective filling of the large pores (approx. 85% of total pore volume).
 |
|
Fig. 41: Saturation adsorption isotherms for Xe (top) and Kr (bottom) in CuIMFU-4l in semilogarithmic scale (left), and corresponding position for Xe (at 170 K) and Kr (at 130 K) atoms in the large and small pores of CuIMFU-4 (right). |
In contrast to Xe adsorption, Kr shows only one adsorption step with a rather usual shape. In the case of Kr adsorption at 130 K, the small pore remains empty even at high pressure (100 kPa, corresponding to 0.5 relative pressure, Figure 41, bottom). This fact looks more surprising taking into account the smaller kinetic radius of Kr atoms and can only be explained by the larger difference between the actual temperature of the measurement (130 K) and the boiling temperature of Kr (120 K) in contrast to Xe (170 K measurement temperature and 165 K boiling temperature).
At the highest loadings in this experiment, the large voids adsorb ca. 55 atoms of Xe and ca. 70 atoms of Kr in 83 available positions, providing a very dense packing of adsorbed noble gas atoms. Strong van der Waals interaction of adsorbed noble gas atoms with framework atoms and also between adsorbed Xe and Kr atoms lead to an apparent increase of the freezing point for Xe and Kr and the formation of a quasi-solid structure of noble gas atoms inside the voids above their boiling temperature – a phenomenon to the best of our knowledge has not been reported before.
The detailed crystallographic study of this quite complicated gas adsorption phenomenon was only possible by using high-resolution synchrotron powder diffraction. Precise determination of crystal structures at different pressures and temperatures allowed accurate interpretation of adsorption isotherms, which is very important for unusual and very rare type of isotherms observed for Xe adsorption.
Principal publication and authors
Formation of a quasi-solid structure by intercalated noble gas atoms in pores of CuI-MFU-4l metal–organic framework, O.V. Magdysyuk (a), D. Denysenko (b), I. Weinrauch (c), D. Volkmer (b), M. Hirscher (c) and R.E. Dinnebier (a), Chem. Commun. 51, 714-717 (2015); doi: 10.1039/C4CC07554D.
(a) Max Planck Institute for Solid State Research, Stuttgart (Germany)
(b) Augsburg University, Augsburg (Germany)
(c) Max Planck Institute for Intelligent Systems, Stuttgart (Germany)
References
[1] A.U. Czaja et al., Chem. Soc. Rev. 38, 1284 (2009).
[2] D. Denysenko et al., Angew. Chem. 126, 5942 (2014).



