- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structural biology
- Elucidating the architecture of transcription factor IIIC
Elucidating the architecture of transcription factor IIIC
The molecular machines that read the messages stored within DNA require the concerted action of transcription factors, molecules that ensure the right messages are read at the correct time. By obtaining a detailed picture of a transcription factor’s atomic structure we can hope to understand how it functions.
In cells, transfer RNAs (tRNAs) play a vital role in the translation of the DNA message into proteins. Transcription of these relatively short RNA molecules is controlled by the proteins of the RNA polymerase III transcription machinery. The genes encoding tRNAs contain two highly conserved promoter sequences called ‘A box’ and ‘B box’, which bind to a protein called transcription factor IIIC (TFIIIC). This flexible, multi-subunit molecule is remarkable in that it can accommodate a great variety of different sized spacing’s between the crucial A and B boxes. The precise nature of how TFIIIC can achieve this is unclear. The protein is hypothesised to be divided into two DNA-binding sub-complexes, known as τA and τB for their A box and B box binding ability, that are joined by a flexible linker.
We have pieced together many of the individual subunits of TFIIIC in previous studies [1-3], with all of the available crystal structures for this protein complex having been solved using beamlines at the ESRF. These valuable structures have provided high-resolution insights into the two DNA-binding sub-complexes of TFIIIC. Despite this, the question of how τA and τB are linked within the structure of TFIIIC has remained a mystery.
Using beamlines ID23-1 and ID23-2, we were able to gain further understanding of the architecture of TFIIIC. Guided by low resolution chemical cross-linking data, we were able to crystallise fragments of the two largest subunits of TFIIIC, τ131 and τ138, which appeared to be critical in forming the linker between the two DNA-binding sub-complexes.
The 1.4 Å crystal structure of τ138 contained an extended winged helix domain, providing the first structural information for this, the largest subunit of TFIIIC. τ138 is predicted to contain several of these domains along its length, and this high-resolution structure will be valuable in modelling the rest of this subunit. However, subsequent biochemical analysis revealed that this particular fragment of TFIIIC was not essential for the linker between τ131 and τ138. We therefore focussed on the τ131 crystal structure, which we could show to be essential for the linker. This crystal structure was of particular interest, as this fragment of TFIIIC is also crucial in assembling other proteins of the RNA polymerase III transcription machinery at tRNA genes.
 |
|
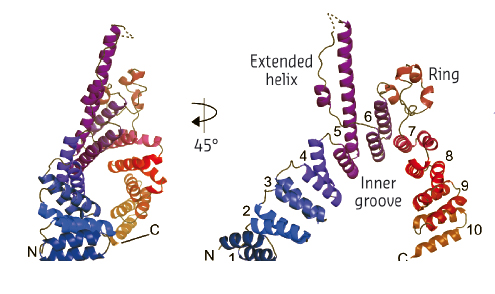
Fig. 125: The TPR array structure of τ131. |
In contrast to the τ138 structure, diffraction from the τ131 crystals was limited to 3.4 Å resolution. Nevertheless, we were able to solve and build the structure (Figure 125). The structure of τ131 reveals an unusual arrangement of several α-helical motifs called tetra-trico-peptide repeats (TPRs). TPRs are ubiquitous in protein complexes, forming extended surfaces over which many other proteins can assemble. However, the structure revealed some additional features, including an extended, flexible helix and a disordered coil that separates the TPR array into two arms (Figure 125). Further biochemical and in vivo analysis using mutants of this ‘TPR array’ have revealed that the τ131 subunit contains many overlapping binding sites for other proteins involved in RNA polymerase III transcription. These experiments, guided by the crystal structure, have led us to propose a model for how TFIIIC might assemble the other proteins required for tRNA transcription.
 |
|
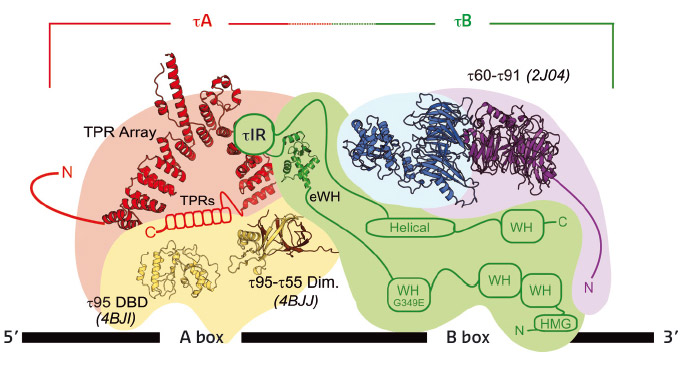
Fig. 126: The current model for the topology of TFIIIC. All of the available crystal structures have been solved at the ESRF. |
The τ131 structure has provided a rational framework with which to address many questions concerning the role of TFIIIC, from how the two DNA-binding sub-complexes are linked, to how TFIIIC interacts with DNA and how TFIIIC assembles the machines required at tRNA genes. The ESRF continues to help us address the structural questions related to this unusual molecule (Figure 126).
Principal publication and authors
Architecture of TFIIIC and its role in RNA polymerase III pre-initiation complex assembly, G. Male (a), A. Von Appen (a), S. Glatt (a), N.M.I. Taylor (a), M. Cristovao (a), H. Groetsch (a), M. Beck (a) and C.W. Müller (a), Nature Communications 6, 7387 (2015); doi: 10.1038/ncomms8387.
(a) European Molecular Biology Laboratory (EMBL), Heidelberg (Germany)
References
[1] N.M. Taylor et al., Nucleic Acids Research 41, 9183-96 (2013).
[2] N.M. Taylor et al., J. Biol. Chem. 288, 15110-15120 (2013).
[3] A. Mylona et al., Molecular Cell 24, 221-32 (2006).



