- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structural biology
- How chemicals can combine to form deadly cocktails from harmless components
How chemicals can combine to form deadly cocktails from harmless components
Endocrine-disrupting chemicals act on nuclear hormone receptors such as the human pregnane X receptor (PXR). Two such chemicals were found to interact with each other in the PXR ligand-binding pocket forming a ‘supramolecular ligand’ that is a more potent PXR activator than either of the two chemicals alone.
Many chemicals released into the environment target nuclear receptors, proteins that control gene expression networks in response to small signalling molecules [1]. These external compounds are classed as endocrine-disrupting chemicals as they are thought to change gene expression patterns, leading to harmful physical effects. While these compounds are generally harmless individually at the doses at which we are exposed, the body is constantly in contact with mixtures of these compounds in the environment. It has long been known that combinations of these chemicals can have additive, antagonistic or synergistic effects, but there has been no understanding of the molecular basis for these effects.
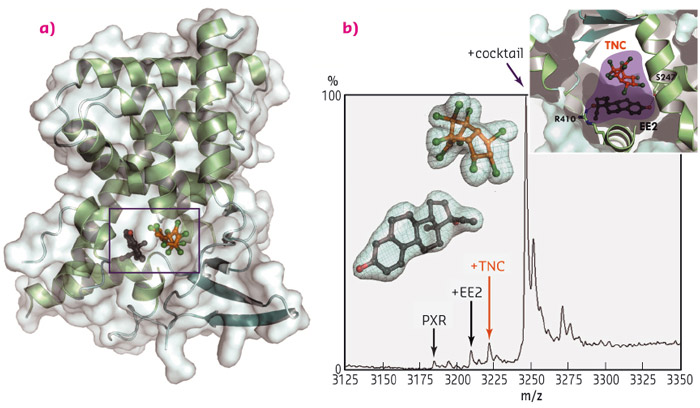
We have used extensive screening methods and biophysical characterisation to identify two endocrine-disrupting chemicals that act synergistically on the human pregnane X receptor (PXR), located mainly in the nucleus of liver, colon and intestinal cells. The two compounds identified are the organochlorine pesticide trans-nonachlor (TNC), and the active ingredient of most contraceptive pills, the synthetic oestrogen derivative 17α-ethinylestradiol (EE2); two compounds with very different uses that are commonly found in our environment. Interestingly, we observed that the two compounds not only bind concomitantly to the ligand-binding pocket of the receptor, but they do so cooperatively, i.e. binding of one molecule promotes high affinity binding of the second. We found that the binary mixture interacts with PXR with a 50- to 100-fold greater affinity than each compound alone, and accordingly, induces a biological response at much lower doses than the individual components. By solving crystal structures of PXR bound to the two chemicals individually and in combination, we have been able to propose a molecular mechanism for the observed synergistic effect between the molecules (diffraction data were collected at beamline ID23-2). The crystal structures show that EE2 and TNC mutually stabilise each other via strong inter-ligand contacts within the ligand-binding pocket to form a so-called ‘supramolecular ligand’ (Figure 124). A large number of interactions with surrounding amino acids allow the supramolecular ligand to tightly hold the binding pocket in the active conformation, causing a sustained activation of the receptor.
 |
|
Fig. 124: Trans-nonachlor (TNC), a pesticide, and 17α-ethinylestradiol (EE2), the active ingredient of the contraceptive pill, combine within the human nuclear receptor PXR to form a supramolecular ligand with enhanced activity. (a) The structure of PXR bound to TNC (orange) and EE2 (dark grey). (b) Electrospray ionisation mass spectrometry under native conditions reveals that EE2 and TNC bind simultaneously to PXR with a much greater affinity than either compound alone. The electron density of the bound chemicals (2Fo-Fc contoured at 1σ) is shown as a mesh. |
These results constitute a proof of concept that opens the way to a wide field of study. There are about 150,000 compounds in our environment that could have unexpected effects on human health through combined action, given their recognised or assumed safety as isolated substances. The results have the potential to change current policies in risk assessment for chemicals and could lead to the development of improved, safer chemicals in the future.
Principal publication and authors
Synergistic activation of human pregnane X receptor by binary cocktails of pharmaceutical and environmental compounds, V. Delfosse (a,b,c), B. Dendele (c,d,e,f), T. Huet (a,b,c), M. Grimaldi (c,d,e,f), A. Boulahtouf (c,d,e,f), S. Gerbal-Chaloin (c,g), B. Beucher (a,h,i), D. Roecklin (j), C. Muller (j), R. Rahmani (k), V. Cavailles (c,d,e,f), M. Daujat-Chavanieu (c,g,l), V. Vivat (j), J-M. Pascussi (c,i,j), P. Balaguer (c,d,e,f) and W. Bourguet (a,b,c), Nature Communications, 6, 8089 (2015); doi: 10.1038/ncomms9089.
(a) Inserm U1054, Montpellier (France)
(b) CNRS UMR5048, Centre de Biochimie Structurale, Montpellier (France)
(c) Université de Montpellier (France)
(d) IRCM, Institut de Recherche en Cancérologie de Montpellier (France)
(e) Inserm, U1194, Montpellier (France)
(f) ICM, Institut régional du Cancer de Montpellier (France)
(g) Inserm U1040, Montpellier (France)
(h) Inserm U661, Montpellier (France)
(i) CNRS UMR5203, Institut de Génomique Fonctionnelle, Montpellier (France)
(j) NovAliX, Illkirch (France)
(k) INRA UMR 1331,TOXALIM, Sophia-Antipolis (France)
(l) CHU de Montpellier, Institut de Recherche en Biothérapie (France)
References
[1] V. Delfosse et al., Acta Pharmacol Sin 36, 88-101 (2015).



