- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Electronic structure, magnetism and dynamics
- Determination of lanthanide-iron cluster interaction in butterfly molecules through combined XMCD and standard magnetometry
Determination of lanthanide-iron cluster interaction in butterfly molecules through combined XMCD and standard magnetometry
Lanthanide organic compounds containing 3d transition elements are promising candidates for the development of single-molecule magnets with a high energy barrier. The goal of this work was to combine the magnetic moment and the relatively strong exchange interactions of 3d transition metals with the anisotropy naturally provided by lanthanide ions.
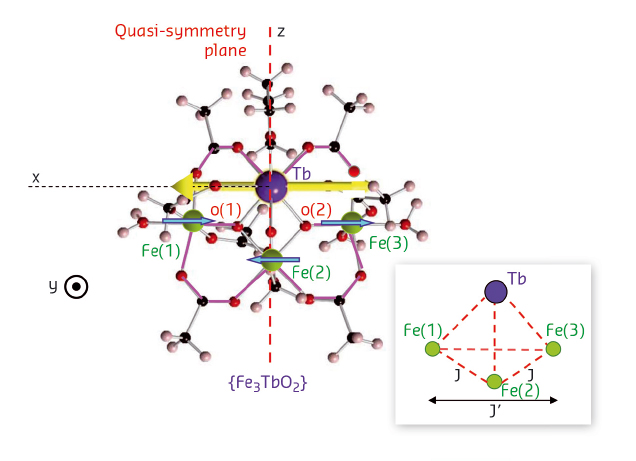
Butterfly molecules {Fe3LnO2} consist of a cluster of three Fe+3 ions forming a triangle (“Fe3”) and a single lanthanide ion Ln+3, all connected by oxygen atoms and surrounded by an organic envelope. The Fe3-Ln cluster shows a butterfly shape (Figure 62). Magnetically, the Fe3 subcluster behaves as a robust entity with total spin 5/2 antiferromagnetically coupled to the Ln magnetic moment [1]. The precise determination of the interaction between the Fe3 cluster and Ln requires their magnetic moments to be measured. Standard methods such as vibrating sample magnetometry (VSM) produce the total moment of the molecule. This allows mFe3 to be obtained from, e.g., the {Fe3YO2} compound, where Y plays the role of a nonmagnetic pseudo rare earth, but in the case of magnetic rare earths, the Fe3 and Ln magnetic moments cannot be discriminated one from the other. In contrast, for an applied magnetic field, the combination of XMCD and VMS makes this separation possible. The results and analysis for the Kramers ions Gd+3 and Dy+3 were reported previously [1], while those of the non-Kramers ions Tb+3 and Ho+3 are highlighted here.
 |
|
Fig. 62: Structure of the {Fe3TbO2} complex. The magnetic moment of Tb (yellow arrow) is perpendicular to the molecule quasi-symmetry plane. Inset: Magnetic cluster formed by the Fe3 unit and the lanthanide. |
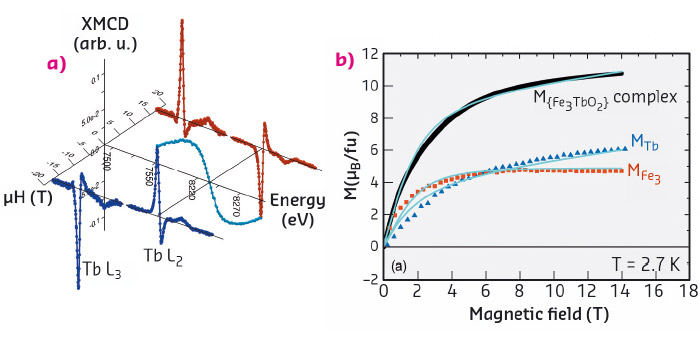
A set of XMCD spectra recorded at the L2,3 edges as a function of the applied magnetic field up to μ0H = 17 T was collected at beamline ID12 at 2.7 K (Tb) and 2.2 K (Ho). Constant temperature M(H) curves plotted in intensity (arbitrary) units were constructed from the spectra (Figure 63a). On the same samples and at the same temperatures, M(H) curves in absolute units were obtained by VMS, as well as for the {Fe3YO2} complex to estimate the contribution of the Fe3 cluster. At saturation, collinearity of the Ln and Fe3 magnetic moments is assured. This allows the determination of the Ln magnetic moment at high fields, which can be used to scale the XMCD magnetisation curve to absolute units. In this way, the MLn(H) curve specific to the lanthanide can be extracted (Figure 63b).
 |
|
Fig. 63: The process to determine interaction constants in {Fe3TbO2}: (a) XMCD spectra and derived M(H) curve in a.u.; (b) Total magnetisation from VSM (black), lanthanide contribution in absolute units from XMCD (blue triangles) and fit within an interaction model (continuous lines). |
A theoretical model for the Fe3-Ln magnetic cluster is proposed with a hamiltonian including crystal field and Zeeman terms, as well as an exchange interaction term between the Fe3 cluster and the lanthanide ion. The Ln moment is modelled through a fictitious ½ spin. While this is straightforward for the groundstate doublet of the Kramers ions case [1], it is only possible for the non-Kramers ions like Tb and Ho under particular conditions. These are well fulfilled at very low temperature, as in the XMCD and VSM measurements, for the two lowest energy singlet states of Tb+3 and roughly fulfilled for Ho+3, according to ab initio calculations. A fit of the calculated MLn(H) curves to the scaled XMCD experimental curve yields to interaction constants along the spin direction JTb-Fe3/kB= −1.5 K and JHo-Fe3/kB= −2.8 K. The evolution of the cluster groundstate with increasing applied field shows a reorientation of the Fe3 cluster magnetic moment from antiparallel to parallel to that of the lanthanide.
The determination of the exchange interaction between transition metal and lanthanide ions in single molecule magnets can be tackled by a combination of XMCD and standard magnetometries to extract magnetic moments, along with a theoretical framework which includes all the relevant terms in the magnetic cluster. The method has been proven in the series of {Fe3LnO2} complexes with Ln = Gd, Dy, Tb and Ho, at very low temperatures, using a fictitious ½ spin to simulate the lowest energy states in both Kramers and non-Kramers ions.
Principal publication and authors
Intracluster interactions in butterfly {Fe3LnO2} molecules with the non-Kramers ions Tb(III) and Ho(III), L. Badía-Romano (a,b), J. Rubín (a,c), F. Bartolomé (a,b), J. Bartolomé (a,b), J. Luzón (a,d), D. Produis (e),C. Turta (e), V. Mereacre (e), F. Wilhelm (f) and A. Rogalev (f). Phys. Rev. B 92, 064411 (2015); doi: 10.1103/PhysRevB.92.064411.
(a) Instituto de Ciencia de Materiales de Aragón, CSIC-Univ. Zaragoza (Spain)
(b) Dept. Fïsica de la Materia Condensada, Univ. Zaragoza (Spain)
(c) Dept. Ciencia de Materiales e Ingeniería Metalúrgica, Univ. Zaragoza (Spain)
(d) Centro Universitario de la Defensa, Academia General Militar, Zaragoza (Spain)
(e) Institute of Chemistry, Academy od Sciences of Moldova, Chisinau (Republic of Moldova)
(f) ESRF
References
[1] L. Badía-Romano et al., Phys. Rev. B 87, 184403 (2013).



