- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Electronic structure, magnetism and dynamics
- Exhaust gas catalysts in action: operando HERFD-XAS and XES for elucidating the mechanism of NOx-removal by ammonia-SCR
Exhaust gas catalysts in action: operando HERFD-XAS and XES for elucidating the mechanism of NOx-removal by ammonia-SCR
Stringent emission legislation for vehicles requires high performance catalysts to reduce pollutant gases. A knowledge-based design and improvement of these catalysts and their implementation in the vehicle can be achieved by understanding the reactions occurring on the catalyst. The interaction of gases with the catalyst was studied under working conditions with complementary X-ray absorption and emission techniques to identify the reaction mechanism.
Optimisation of combustion engines combined with modern catalytic converters has contributed to a significant reduction of pollutant emissions [1,2]. Recently, new catalysts for diesel cars have been introduced to decrease NOx-emissions. Cu-based zeolites are the active component for the selective catalytic reduction (SCR) of NOx using NH3, which is obtained by decomposition of urea (AdBlue®). There is a debate in the literature on the structure and position of active Cu-sites within the zeolite. A profound understanding of the catalyst operation will support rational catalyst design and the development of control algorithms for exhaust gas aftertreatment systems. Characterisation of the material before and after the reaction can provide initial ideas on the catalyst’s behaviour. However, for comprehensive characterisation, it is essential to study the catalyst under working conditions since many intermediate steps are only observable while the reaction is occurring because the structure of catalysts is dynamic.
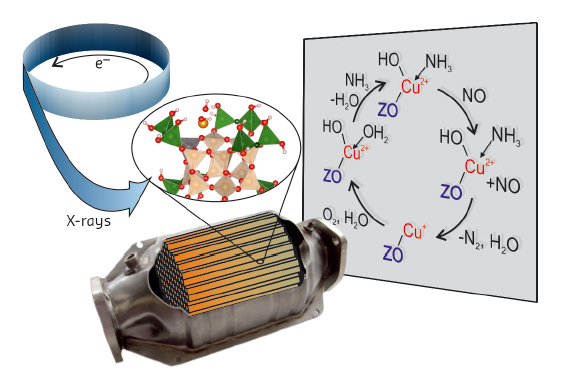
As indicated in Figure 52, X-rays offer the possibility to monitor the catalyst under operating conditions. In addition, they can be used to selectively probe one element, allowing visualisation of the interaction of gas molecules with these sites while ignoring adsorption on the catalyst support material. Using high-energy-resolution fluorescence-detected (HERFD) X-ray absorption spectroscopy (XAS), we could overcome the limitations of standard XAS, leading to detailed measurement of the Cu K edge that contains information on the oxidation state and coordination of the metal sites. Additional valence-to-core X-ray emission spectroscopy (v2c-XES) made identification of adsorbed species at these metal sites possible (e.g. NOx, NH3, H2O), while distinguishing between atoms with close atomic numbers such as O and N.
 |
|
Fig. 52: Strategy of the study of automotive catalysts using X-rays and the suggested mechanism. |
Here we demonstrate the application of these techniques in order to characterise the catalytic sites of Cu-SSZ-13 under reaction conditions. A quartz capillary microreactor was used to mimic a plug-flow reactor as closely as possible. The sample was heated to a realistic reaction temperature and the gas composition analysed at the reactor outlet. To understand the SCR-reaction, model gas conditions were tested in addition to the SCR mixture, allowing the influence of each gas component and interactions between the gases to be studied separately.
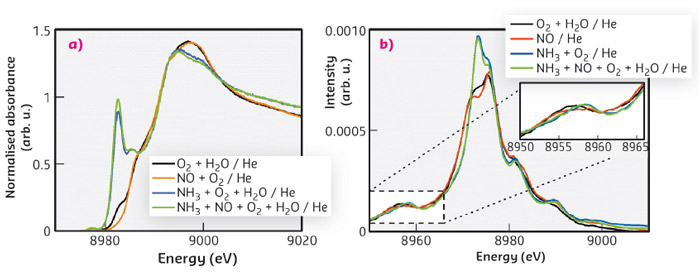
We were able to follow small spectroscopic changes in the presence of different gases via HERFD-XAS and v2c-XES at beamline ID26 (Figure 53). Cu oxidation state and geometry could be quantified by using the pre-edge feature (e.g. Cu2+, 1s2 → 3d9). A more intense feature in the rising edge region is related to Cu+ and becomes even more pronounced for linear coordination of Cu. As Figure 53a shows, this feature is clearly visible in the presence of NH3, indicating reduction and change in coordination at the Cu-site. A decrease of the pre-edge feature under SCR-reaction conditions shows that the mechanism involves a redox step, with slow reoxidation of Cu-sites. XES gives complementary information on the electronic structure which is characteristic for coordinating ligands/adsorbates at the Cu-sites. This was substantiated by DFT calculations.
 |
|
Fig. 53: (a) HERFD-XANES and (b) valence-to-core XES of Cu-SSZ-13 under selected gas compositions at 200°C. |
The small satellite feature Kß” is a valence to core transition from a molecular orbital, based on the interaction of metal orbitals with ligand 2s orbitals. As the energy of the ligand 2s orbital mainly depends on the element present, the position of the Kß” feature acts as a fingerprint for the coordinating atom and can distinguish between the ligand bonding mode of nitrogen or oxygen. The adsorption of NH3 shifted this feature to higher energies, indicating direct coordination via N at the Cu-site (Figure 53b). In the presence of water and under reaction conditions, this feature is broadened to lower energies due to competitive adsorption of NH3 and H2O at the active sites. Weak adsorption of NO could only be observed under NO in He without any additional gases, suggesting weak interaction with the Cu-sites, or reaction from the gas phase with NH3.
In contrast to these results, we observed adsorption of NO via a triple-coordinated oxygen atom for the iron-based SCR-catalyst Fe-ZSM-5 [3].
Principal publication and authors
Structural snapshots of the SCR reaction mechanism on Cu-SSZ-13, T. Günter (a), H.W.P. Carvalho (a), D.E. Doronkin (a), T. Sheppard (a), P. Glatzel (b), A.J. Atkins (a), J. Rudolph (c), C.R. Jacob (c), M. Casapu (a) and J.-D. Grunwaldt (a), Chem. Commun. 51, 9227-9230 (2015). doi: 10.1039/c5cc01758k.
(a) Karlsruhe Institute of Technology (Germany)
(b) ESRF
(c) TU Braunschweig (Germany)
References
[1] E.S.J. Lox in Handbook of Heterogeneous Catalysis, Vol 5, p 2274, Wiley-VCH (2008).
[2] O. Deutschmann, J.-D. Grunwaldt, Chem. Ing. Tech. 85, 595-617 (2013).
[3] A. Boubnov et al., J. Am. Chem. Soc. 136, 13006-13015 (2014).



