- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Complex systems and biomedical sciences
- Two-step nucleation in protein crystallisation revealed by real-time SAXS
Two-step nucleation in protein crystallisation revealed by real-time SAXS
How critical nuclei gain the density and structure from a supersaturated state is still a mystery. By following the kinetics of protein crystallisation using real-time SAXS, it is demonstrated here that nucleation can follow a step-by-step mechanism, i.e. proteins first form disordered aggregates, and then the structure develops inside.
According to the classical nucleation theory, nucleation starts as solute molecules reversibly aggregate in supersaturated solutions and form nuclei with the exact density and structure of the crystals in the final stage. While classical nucleation theory provides a successful description in many systems, studies on the crystallisation of proteins and colloids as well as on biomineralisation have shown features beyond the classical view. A two-step nucleation mechanism has been proposed by ten Wolde and Frenkel [1], where the two order parameters (density and structure) develop sequentially instead of simultaneously, i.e. a metastable intermediate phase (MIP) forms first from the supersaturated solution, and nucleation occurs afterwards inside the MIP (Figure 85a). Despite the existing theoretical concepts, clear experimental evidence and suitable methods to study the early stage of nucleation are rather rare - in particular, when aiming for an ensemble average of the rare-event nucleation process of a real system.
In our work, we studied the crystallisation of b-lactoglobulin (BLG) in solutions in the presence of the divalent salt cadmium chloride (CdCl2). This system has a pseudo-reentrant condensation phase behaviour [2,3]. Interestingly, near the second transition zone, protein aggregates form before crystallisation and are consumed later by crystal growth. Such a system provides ideal conditions for identification of the pathway of crystal nucleation.
 |
|
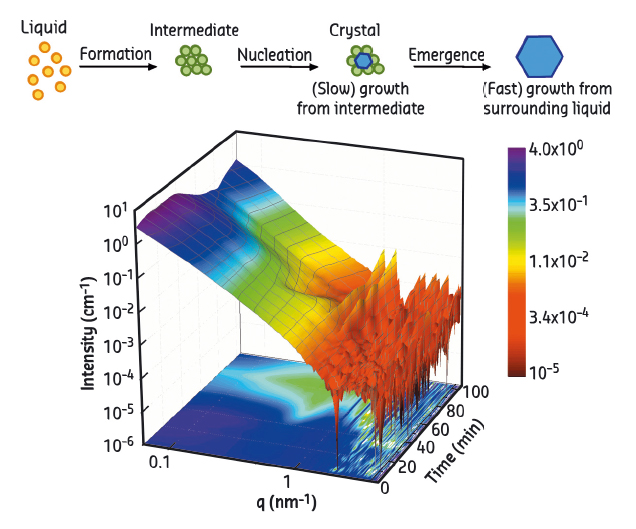
Fig. 85: (a) Schematic of the two-step nucleation mechanism. (b) 3D surface illustration of typical real-time SAXS data. |
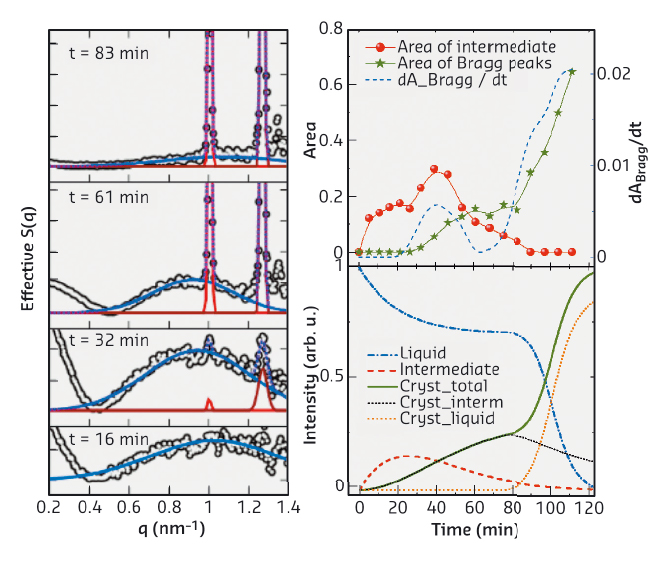
Benefiting from its high resolution in time and space, real-time small-angle X-ray scattering (SAXS) was employed to follow the early stage of crystallisation. SAXS measurements were performed at beamline ID02. A typical real-time SAXS profile from a sample with 20 mg/ml BLG and 15 mM CdCl2 is shown in Figure 85b. The first curve (t = 0) already indicates the formation of protein aggregates. With increasing time, a broad peak at intermediate q-values appears. This peak increases in intensity until Bragg peaks start to appear, with the two most pronounced peaks located at 1.01 and 1.27 nm–1 overlapping with the broad maximum. The Bragg peaks increase in intensity with time, whereas the broad peak shrinks, and eventually disappears. To quantify the relationship between the aggregates and crystals, we evaluated the area under the broad peak and under the two main Bragg peaks. The overall crystallisation kinetics can be described by plotting these parameters as a function of time (Figure 86). The amount of MIP and crystalline phases are labeled in red and green, respectively. The blue curve is the first derivative of the green curve, representing the crystallisation rate. First, the maximum in the green curve coincides with a local maximum in the crystallisation rate, indicating that the crystallisation rate is proportional to the amount of MIP. Second, when the MIP is nearly consumed (~ 80 min), the fraction of the crystalline phase reaches a plateau value. After that, it increases again, even more rapidly.
 |
|
Fig. 86: Analysis of real-time SAXS data and identification of the two-step nucleation mechanism. |
From the crystallisation kinetics obtained from time-resolved SAXS, we can identify the kinetic signature of a two-step nucleation process in the early stage of protein crystallisation. The first regime of the kinetics is dominated by a two-step nucleation and growth within the MIP, i.e. protein aggregates are formed as a MIP, followed by the nucleation of crystals within the MIP. During this period, the number of crystals increases with time, but the growth rate is slow due to the low mobility in the surrounding aggregates. The second regime of the kinetics is dominated by the growth in the dilute phase: the consumption of the MIP leads to the exposure of the crystals to the free molecules in the dilute phase, which accelerates the crystal growth. This real-time study not only provides evidence for a two-step nucleation process for protein crystallisation, but also elucidates the role and the structural signature of the metastable intermediate phase in this process.
Principal publication and authors
Real-time observation of nonclassical protein crystallization kinetics, A. Sauter (a), F. Roosen-Runge (b), F. Zhang (a), G. Lotze (c), R.M.J. Jacobs (d) and F. Schreiber (a). J. Am. Chem. Soc. 137, 1485-1491 (2015); doi: 10.1021/ja510533x.
(a) Institut für Angewandte Physik, Universität Tübingen (Germany)
(b) ILL, Grenoble (France)
(c) ESRF
(d) Department of Chemistry, CRL, University of Oxford (UK)
References
[1] P.R. ten Wolde and D. Frenkel, Science 277, 1975-1978 (1997).
[2] F. Zhang et al., Phys. Rev. Lett. 101, 148101 (2008).
[3] A. Sauter et al., Faraday Discuss. 179, 41-58 (2015).



