- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- X-ray Imaging
- Coarsening and break-up revealed by X-ray microtomography of phase-separated glasses
Coarsening and break-up revealed by X-ray microtomography of phase-separated glasses
In their molten state, some silicate glasses spontaneously phase-separate: they form domains of different compositions [1, 2]. Such domains can take different shapes, such as droplets or interconnected channels. The physics is then very similar to an emulsion of oil and water, with the microstructure gradually coarsening to reduce the interfacial energy. In the case of a silicate glass, the liquid state is obtained at temperatures generally higher than 700°C, and by cooling it below this temperature one can “freeze” the system. The domains formed in the liquid state then become a microstructure in the solid, and can be used to tune some physical properties. Using X-ray tomography at ID19, we were able to visualise the formation of this microstructure in a barium borosilicate glass in three dimensions (3D), and observe the effect of the time spent in liquid state.
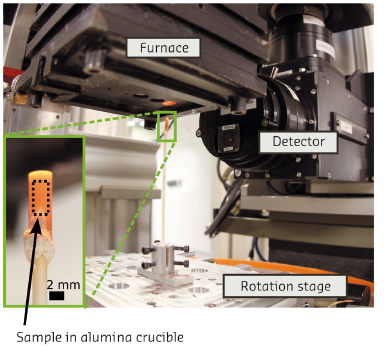
With X-ray tomography, one reconstructs 3D images of the absorption of a sample from several hundreds of radiographs. The high intensity flux at ID19, together with a setup suited for ultrafast acquisition, allows fast scans. Moreover, a dedicated furnace is used such that the same sample can be heated for some time at a given temperature then quenched to be scanned, then heated again, and so on (Figure 66).
 |
|
Fig. 66: Experimental setup at ID19. The sample, a 2 mm cylinder, is placed in an alumina crucible mounted on a rotation stage. A furnace, mounted on a vertical translation stage, is lowered over the sample for the heat treatments. In this image the X-rays would be arriving from the left. |
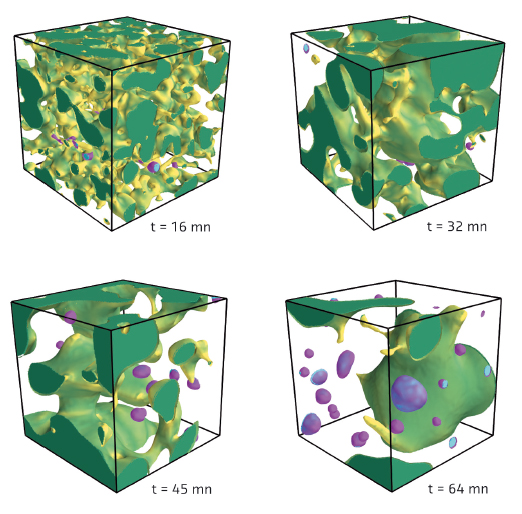
After image processing, we were able to observe the intricate geometry of one of the two phases (Figure 67). At the beginning of the heat treatment, the system is bicontinuous, which means that there is only one domain for each phase, and these domains are interpenetrating. The geometry is similar to a sponge, where there is only one “solid” part, that is contiguous (otherwise it would collapse), and only one “void” part (otherwise the water could not fill it all). In fact, it is possible to produce a “glass sponge” by leaching one of the two separated phases with acid, which, for example, is used to produce porous glass membranes.
 |
|
Fig. 67: 3D rendering of the barium-rich phase during coarsening. The largest domain, in green, is percolating. Other domains, in purple, are isolated droplets. The shades of colour represent the mean curvature value. The lateral size of the cubes is 140 µm. |
During the heat treatment, we were able to measure how the typical size of the domains increases in order to reduce the interfacial area where the two immiscible phases are in contact. General predictions suggest that this coarsening should be self-similar. This means that the geometry of the domains is statistically the same at any time provided that all lengths are divided by a characteristic length scale, which itself grows with time. The experiments confirmed this theoretical prediction, up to a limit: the coarsening is slowed down by the fragmentation of the less viscous phase into disconnected droplets. This important fragmentation mechanism was identified for the first time thanks to the possibility of following the same sample at different heat treatment times.
The experimental setup at ID19 has recently been improved with a new camera and rotation stage. The acquisition time has been reduced from a few minutes to a few seconds to obtain 3D images with micrometric resolution. This has triggered new studies, which are ongoing, to investigate the origin of the unexpected fragmentation. It is now possible to follow in situ the coarsening (without quenching to room temperature). In particular, the dynamics of evolution of domain shapes are considered, and point towards hydrodynamics effects. Such understanding of microstructure formation could lead to new ways of building tailored microstructures in phase-separated glasses.
Principal publication and authors
D. Bouttes (a), E. Gouillart (b), E. Boller (c), D. Dalmas (b) and D. Vandembroucq (a), Physical Review Letters, 112, 245701 (2014).
(a) Laboratoire PMMH, UMR 7636 CNRS/ESPCI/University Paris 6 UPMC/University Paris 7 Diderot (France)
(b) Surface du Verre et Interfaces, UMR125 CNRS/Saint-Gobain, Aubervilliers (France)
(c) ESRF
References
[1] A.J. Bray, Theory of phase-ordering kinetics. Advances in Physics, 43(3), 357-459 (1994).
[2] O.V. Mazurin and E.A. Porai-Koshits (Eds.). Phase separation in glass, Elsevier (1984).



