- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Structure of materials
- Illuminating the physical process of catalyst deactivation
Illuminating the physical process of catalyst deactivation
Synchrotron-based X-ray diffraction computed tomography (XRD-CT) is a technique developed at the ESRF for the purpose of revealing detailed chemical compositional information in solid materials in 2 and 3 spatial dimensions [1]. Much of this ‘chemical imaging’ has focused on the study of pre-shaped catalyst bodies often as a function of time. Here we demonstrate how the technique has been used to understand the importance of spatial variation during the process of sulfur-induced solid-state deactivation of low temperature Cu/ZnO/Al2O3 water-gas shift catalysts.
Catalyst deactivation by sulfur poisoning is a common problem for a wide range of catalytic processes and which is likely to become more problematic as the chemical industry moves from relatively clean feeds, such as natural gas and crude oil, to alternative feedstocks, such as bitumen tars and some forms of biomass. Cu/ZnO catalysts represent the industrial standard for the production of methanol and the water–gas shift (WGS) reaction. The catalysts are however very susceptible to sulfur poisoning (ZnO for example is often used as a ‘sulfur scrubber’). However the process by which sulfur poisons these catalysts is still up for debate. It is thought that sulfur, in the form of either H2S or similar compounds, accumulates on the surface of the metallic Cu species blocking the active sites or else induces a phase transformation of the Cu and ZnO into the respective sulfides and oxy-sulfides. Since catalysts are used in pre-shaped forms, i.e. as millimetre-sized catalyst bodies, to understand deactivation in real catalyst samples needs consideration of the spatial perspective in order to assess macroscopic effects leading to catalyst deactivation. XRD-CT is ideal for assessing crystalline phase distribution at the µm scale, whereas XAFS/XRD mapping can be used to determine the relative importance of poisoning via sulfur adsorption vs. solid-state transformation of the active Cu/ZnO phase(s) into sulfide phases.
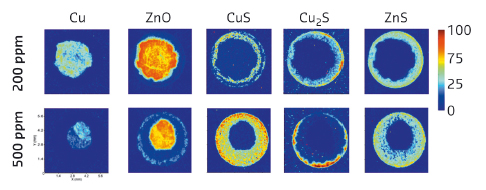
Sulfur, in the form of H2S, was added to the catalytic feed during the WGS reaction and the results of the XRD-CT data recorded (at beamline ID15B, now ID31) as a function of increasing sulfur concentration are given in Figure 144. The initial crystallline Cu and ZnO phases disappear with increasing [H2S] in the feed and both phases lose intensity and ‘shrink’ towards the core of the body. New sulfide phases evolve on the outside, while the increasing [H2S] leads to a thicker sulfide shell. Consequently, the thicker the sulfide shell, the greater the loss in catalytic activity. In the sample poisoned using 500 ppm H2S, over 80% of the cylinder volume is converted into sulfide-containing phases and almost all catalytic activity is lost.
 |
|
Fig. 144: 2D XRD intensity colour maps for various crystalline phases (top) after two concentrations of H2S (left) were introduced in the WGS reactant feed. Right – colour scale map |
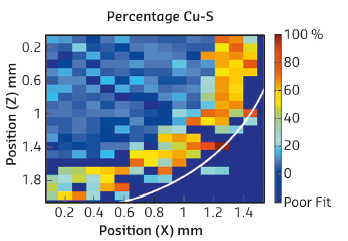
Whilst XRD-CT is able to identify the distribution of crystalline components over the 2D cross-section, it is insensitive to the presence of small amounts of adsorbed sulfur species on the surface of metallic Cu, which may block the active site. For this purpose, a chemical map derived from the fitting of the Cu-NN (nearest neighbour) contribution from XAFS spectroscopy on a section of the microtomed catalyst is given in Figure 145. The main Cu nearest neighbour component is < 2 Å at the core and > 2.1 Å towards the edge, consistent with the presence of CuO at the core and CuS at the edge. There was no clear evidence for the presence of either Cu-S or Zn-S phases in the centre of the sample [2].
 |
|
Fig. 145: Derived composition data from the Cu-‘O’ bond distance map of a microtomed cross-section of a Cu/ZnO/Al2O2 catalyst body treated with 200 ppm H2S. Right – colour scale map |
The formation of sulfide phases at the sample edges is highly detrimental to the catalytic activity of the sample since the catalyst deactivates before all Cu/ZnO has been lost. Although industrial Cu/ZnO/Al2O2 catalysts are unlikely to see anything like the amount of H2S used in this study, the tendency for Cu and Zn to adsorb S does mean that, over time, sulfide phases will accrue in much the same way as observed here. Clearly it is important to consider the spatial component when trying to determine the structure-activity relationships in real heterogeneous catalyst samples with imaging methods critical for such purposes.
Principal publication and authors
A.M. Beale (a,b,c), E.K. Gibson (b,c), M.G. O’Brien (a), S.D.M. Jacques (c,d), R.J. Cernik (d), M. di Michiel (e), P.B. Cobden (f), O. Pirgon-Galin (f), L. van de Water (g), M.J. Watson (g) and B.M. Weckhuysen (a), J. Catal. 314, 94 (2014).
(a) Inorganic Chemistry and Catalysis, Debye Institute for Nanomaterials Science, Utrecht University (The Netherlands)
(b) Department of Chemistry, University College London (UK)
(c) UK Catalysis Hub, Research Complex at Harwell, Rutherford Appleton Laboratory, Didcot (UK)
(d) School of Materials, University of Manchester (UK)
(e) ESRF
(f) Energy Research Centre of the Netherlands (ECN), Petten (The Netherlands)
(g) Johnson Matthey Technology Centre, Billingham (UK)
References
[1] A.M. Beale, S.D.M. Jacques, E.K. Gibson and M. Di Michiel, Coord. Chem. Rev. 277, 208 (2014).
[2] A.M. Beale, E.K. Gibson, M.G. O’Brien, S.D.M. Jacques, R.J. Cernik, M. di Michiel, P.B. Cobden, O. Pirgon-Galin, L. van de Water, M.J. Watson and B.M. Weckhuysen, J. Catal. 314, 94 (2014).



