- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Soft condensed matter
- Solvent effects promote triple helix formation in amphiphilic discotics
Solvent effects promote triple helix formation in amphiphilic discotics
Self-assembling synthetic molecules in water is of great interest for the development of functional soft matter and finds applications in regenerative medicine and sensors. Defining rules for effective self-assembly in water with the ability to control the shape and function of the supramolecular structures formed is, however, less trivial. A delicate balance between hydrophobic effects and other non-covalent interactions is often required but not easy to achieve.
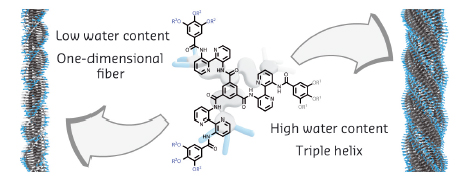 |
|
Fig. 37: Self-assembly of an amphiphilic discotic into (left) one-dimensional helical fibres at low water content and (right) triple helical structures at high water content. |
With the help of small-angle X-ray scattering (SAXS) at beamline ID02, in combination with circular dichroism (CD) spectroscopy and cryogenic transmission electron microscopy (cryo-TEM), we have now unravelled how the self-assembly behaviour in water of an amphiphilic discotic molecule (Figure 37, middle) is affected by the presence of isopropanol (IPA), added to enhance its solubility. The molecular structure of the discotic amphiphile is designed to form bundled structures in H2O (Figure 37, right) and incorporates both hydrophilic/hydrophobic peripheral side chains and a core that strongly aggregates via π-π interactions. In pure IPA, the hydrophobic effect is absent and only π-π interactions are operative, resulting in one-dimensional helical fibres (Figure 37, left).
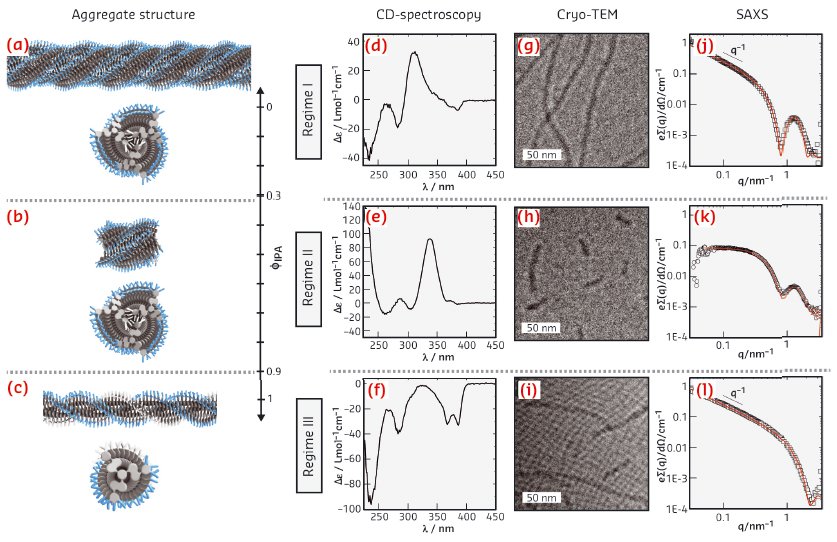 |
|
Fig. 38: a-c) Schematic depiction of the three regimes in self-assembly of the discotic amphiphile in IPA–H2O. CD-spectra of c = 3x10-4 M and T = 0°C for: d) fIPA = 0.25; e) fIPA = 0.5; f) fIPA = 1; Cryo-TEM images at c = 3x10-4 M for g) fIPA = 0.25; ) fIPA = 0.75; i) fIPA = 1; SAXS profiles (symbols) and form factor fits (lines) in IPA–H2O mixtures at c = 3×10-4 M and T = 20°C for: j) fIPA = 0.25; k) fIPA = 0.5; l) fIPA = 1. |
The behaviour of the discotic amphiphile was investigated as a function of solvent composition in IPA–H2O mixtures and temperature. Remarkably, three distinct structural regimes were detected with all techniques (Figure 38). SAXS showed that a 1D helical fibre was present at high IPA concentration, evidenced by a scattering curve typical for long elongated objects with a homogenous electron length density (regime III). The addition of water induced the formation of small, bundled aggregates, showing a signature scattering curve for core–shell assemblies with a low aspect ratio (regime II). At high water content, long, one-dimensional core–shell assemblies are formed, consisting of 3-4 fibres in the cross-section. Because the discotics have a flexible propeller shape and are never completely flat, the most likely conformation is a triple helix of helices. The results obtained by SAXS measurements were corroborated by cryo-TEM measurements: short fibres were observed for regime II and long fibres for regimes I and III. CD spectroscopy showed strong Cotton effects in all three regimes, confirming the presence of helical aggregates.
The most noteworthy finding of this research is that the boundaries of the three regimes coincide with the two extrema (Φmax and Φmin) that are present in the excess enthalpy of mixing phase diagram of IPA–H2O mixtures [1]. This suggests that the solvent mixing thermodynamics drive the observed morphological transitions, as also suggested by Trappe and co-workers for the phase behaviour of poly(N-isopropylacrylamide) in IPA–H2O mixtures [2]. To confirm that the boundary between regime II and III is indeed coupled to excess enthalpy of mixing, we performed a temperature-dependent experiment at ΦIPA= 0.95. At 80°C, Φmax is well below 0.95 while at 0°C Φmax shifts to values larger than 0.95. This means that cooling a solution of the discotic amphiphile in ΦIPA = 0.95 should induce a change in the nature of the aggregates typical for regime III to an aggregate typical for regime II. Indeed, the CD spectrum at 10°C is typical for regime III, while it is typical for regime II at 0°C.
The above findings demonstrate that the self-assembly of the discotic amphiphile discussed here in IPA–H2O mixtures is intricately linked to the mixing behaviour of the IPA–H2O solvent mixture. This finding is highly important since the nanostructures obtained upon self-assembly in water can be greatly influenced by the preparation method. Frequently, samples are prepared by the direct injection into water of solutions of small self-assembling units in a “good” solvent such as an alcohol or THF. Understanding how the solvent composition influences the aggregate structure helps us to unravel the pathways by which the final structures are formed in water.
Principal publication and authors
M.A.J. Gillissen (a,b), M.M.E. Koenigs (a,b), A.J.H. Spiering (a), J.A.J.M. Vekemans (a), A.R.A. Palmans (a,b), I.K. Voets (a,b) and E.W. Meijer (a,b), J. Am. Chem. Soc. 136, 336-343 (2014).
(a) Laboratory of Macromolecular and Organic Chemistry, TU Eindhoven (The Netherlands)
(b) Institute of Complex Molecular Systems, TU Eindhoven (The Netherlands)
References
[1] a) F. Franks and D.J.G. Ives, Q. Rev. Chem. Soc. 20, 1 (1966); b) T. Katayama, Kagaku Kogaku Ronbun. 26, 361 (1962); c) R.F. Lama, B.C.Y. Lu, J. Chem. Eng. Data 10, 216 (1965).
[2] I. Bischofberger, D.C.E. Calzolari, P. De Los Rios, I. Jelezarov and V. Trappe, Scientific Reports 4, 4377 (2014).



